NIR-Based Chemical Imaging As An Anticounterfeiting Tool
Counterfeit drugs pose a significant and fastgrowing threat to public health and to the pharmaceutical industry. For an individual patient, treatment with a counterfeit drug is at best ineffective, and, at worst, lethal. For pharmaceutical manufacturers, both reputation and revenues are at stake.
Near infrared (NIR) chemical imaging-as distinct from NIR spectroscopy-is a powerful analytical technique that is being used increasingly in the fight against counterfeit drugs. Capable of simultaneously analyzing a number of tablets or capsules, NIR imaging can be automated to provide rapid genuine/ fake detection. At higher magnifications, it delivers detailed information about the abundance and location of chemical species. Here we consider the challenges posed by counterfeit pharmaceuticals and the unique attributes of NIR that make it such an important tool in dealing with them.
Identifying Counterfeits
Counterfeit drugs can take many forms: they may contain no active pharmaceutical ingredient (API); they may have the wrong API; or they may be made using the correct API but in the wrong concentration or form. While intuitively it might appear that those containing the correct API should present the least danger, this is not necessarily the case.
Tablets, for example, are engineered in a highly sophisticated manner in order to ensure optimal in vivo breakdown and dose delivery. Even a counterfeit tablet containing API at the correct concentration may fail to replicate the right delivery action, releasing API at an incorrect rate, at the wrong time or in the wrong place, thus compromising efficacy. Initial detection of counterfeits is often through their packaging, with covert markers and holograms now widely used to identify the genuine product.
Once a counterfeit is uncovered, the potential for harm is assessed in the laboratory. Traditionally the analytical techniques applied are analogous to those used for QA/ QC of the genuine item. They include chromatographic assays which are used to determine what is in the product and at what concentration, and laborious dissolution testing. In practice, the analytical burden associated with counterfeit detection can be high. NIR imaging is a relatively new technique that is generating significant interest within the pharmaceutical industry for this and other applications. Unlike conventional NIR spectroscopy, which provides averaged compositional data, NIR imaging gathers spectra for thousands of pixels across the face of a sample, producing spatially resolved data.
At low magnifications, true NIR imaging systems based on two-dimensional detectors (as opposed to linear mapping systems that acquire data one line at a time) have large fields of view, allowing simultaneous analysis of, say, a complete blister pack or a composite selection of tablets from different sources. This ability to have both real and suspect product in the same field of view means that the identification of counterfeit product can be made without building complex calibration models. Short analysis times of just a few minutes, no requirement for sample preparation, easy automation and minimal manual input, all add to the attractiveness of the technique for rapid and effective counterfeit detection.
Understanding NIR Imaging
Conventional NIR spectroscopy relies on the absorption of light in the wavelength range 700 to 2500 nm to detect the presence of different chemical bonds, and hence species. NIR imaging combines this capability with the spatial resolution capabilities of chemical imaging, not only detecting the presence of chemical species but also pinpointing their location. In place of a single spectrum, NIR imaging simultaneously generates tens of thousands of spectra, each relating to a specific area of the sample. Powerful statistical analysis tools extract pertinent information from the resulting dataset.
In the instrumentation, quartz halogen lamps offer a safe and easily configurable source of illumination, while a liquid crystal tunable filter provides a simple mechanism for wavelength discrimination, maintaining excellent image quality. Spectra are captured using a two-dimensional array, a critical component in a true NIR imaging analyzer. This array eliminates any need to move samples during analysis, accelerating measurement and, importantly, permitting the development of systems that have no moving parts.
Simple optics allow easy configuration of the system for the study of a small area, an individual particle or granule, for example, or a larger region, such as a composite sample of tens of tablets. NIR imaging systems can collect more than 80,000 spectra in minutes, with user-friendly software processing raw data into usable information. Image generation allows a qualitative overview of a sample, which can be useful for making rapid comparisons.
If simple information is required, for example, conformation that a sample is genuine, then this process can be further accelerated by focusing on just a few key wavelengths, slashing data collection and processing times. More detailed quantitative analysis generates objective answers to more challenging questions. Both approaches are important for counterfeit detection, as the following study illustrates.
Investigating A Sample of Anti-Malarial Tablets
Thirty anti-malarial tablets, all white cylinders scored on one side and debossed with a trade name on the other, were imaged using the Sapphire near infrared chemical imaging system. Of these, 10 were the genuine product, while seven of the counterfeits contained paracetamol (acetaminophen) and 13 an unknown, substitute API.
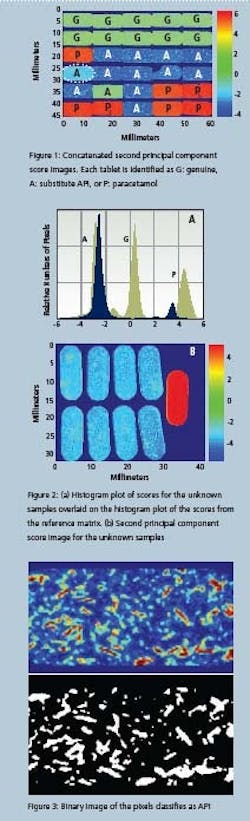
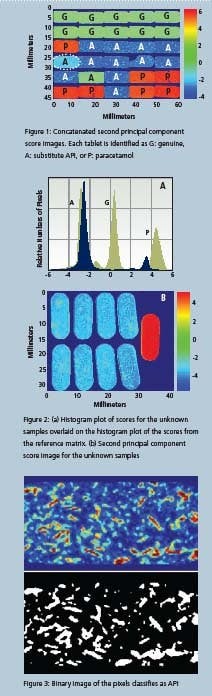
The tablets were analyzed without any sample preparation using a magnification of 40 microns/pixel, with an analysis time of around three minutes for each sample. Initially, a principal component analysis (PCA) was used to extract information from the resulting dataset. PCA is an unsupervised multivariate analysis that essentially extracts distinguishing features from a dataset regardless of their origin (physical or chemical) [1]. It is a useful initial screening tool as can be seen from Figure 1. In this figure, the second principal component score image for each sample is stitched into an array, so that all of the samples can be viewed in one image, even though the data for each of these samples was acquired separately.
The tablets can be classified as: genuine (G); containing paracetamol (P); or containing a substitute API (A), on the basis of the first three principal component scores images. The fourth principal component scores image further differentiates the tablet in row 4, column 1 from all the others. Figure 3: Binary image of the pixels classifi es as API is tablet also exhibits a difference in the second principal component score image. Comparing the data with spectra from a library of known substances identifies the differentiating component as sucrose. This tablet contains more sucrose than any of the others, probably in the coating, as it is evident across the entire sample.
The tablet in row 5, column 2 is correctly identified as genuine despite being wrongly labelled as containing a substitute API. No reference data were used to extract this information. While the color plot is a useful visual image, much additional information can be inferred from a study of the histograms of the scores values.
Figure 3 shows these distributions in grey, for all 30 tablets. Different types of tablets appear to cluster into distinct populations, a finding confirmed by correlating these distributions with spatial location. Other information, such as the standard deviation of the populations provides objective measures of heterogeneity. Common statistical parameters such as skew and kurtosis values, determined by the number of exceedingly low or high value pixels, and the overall shape of the distribution, respectively characterize component localization.
By overlaying histogram values of unknown samples onto these populations, the unknowns can be assigned to a particular class of sample. To illustrate this approach, nine unknown samples were imaged using a large field of view magnification, so that all of the sample data were acquired at the same time, instead of sequentially, as in the first example. The color image of the unknown tablets shows that eight appear identical, while the ninth belongs to a different class.
To determine which class these samples belong to, the associated histogram of their score values is superimposed over that of the original data in Figure 2. The majority of samples overlap with the A population indicating substitute API. The ninth tablet however, has a distribution that most closely resembles the paracetamol class. One of the unique capabilities of NIR imaging is its ability to perform a detailed analysis on the spatial distribution of sample components.
To explore the information available from this type of analysis, all samples with substitute API from the first example 11 in total - were subject to a detailed morphological analysis. In this case, a Partial Least Squares (PLS) algorithm was applied. In contrast to a PCA analysis which requires no user input, PLS is a supervised method in which a library of pure components is used for advanced analysis [1]. Examination of the sample data led to the conclusion that tablets could reasonably be treated as two-component systems consisting of API and excipient, where the excipient class is really a mixture of multiple excipients.
Because there are no pure components available for this study, a library was created from spectra contained within the sample data, using 2220 nm as the marker band for the API and 2250 nm for the excipient mixture. The images in Figure 3 provide information about the distribution of API within the tablet, extracted from the resulting data. The black and white image is derived from the color image by defining threshold values to classify areas as either API or excipient.
Of the 11 tablets analyzed in this manner, three were found to contain a large number of domains of average size. Of the remaining eight samples, four contained domains at the larger end of the observed scale, and the final four had API-rich domains that were relatively small and much less numerous. This difference was significant in comparison with the other eight tablets, and leads to the conclusion that the API is more effectively blended in this tablet group. So it can be concluded that the groups of tablets are produced either in different processes or in a single process with relatively poor control.
NIR imagings ability to identify fake tablets rapidly and detect subtle differences between different samples is clear. NIR imaging data provides information that is important not only in determining the provenance of counterfeit drugs but also for assessing process performance. NIR imaging has developed significantly over the past decade, to the point where rugged, industrial systems, designed with automation in mind, are now available commercially.
Its ability to simultaneously image a number of tablets allows the use of a genuine sample for in situ calibration, allowing rapid and conclusive differentiation between real and counterfeit tablets. With a streamlined analytical method, focusing on just a few key wavelengths, counterfeit detection can be complete in seconds. Automation of the complete procedure ensures repeatability and provides a push-button option for real-time measurement.
At higher magnifications, these same systems can permit more detailed analysis, giving information about the location of chemical species as well as their abundance. In the analysis of counterfeits this gives real insight into a product its provenance and manufacturing history.
References
[1] J Dubois et al. NIR Chemical Imaging for Counterfeit Pharmaceutical Products Analysis. Spectroscopy February 2007.