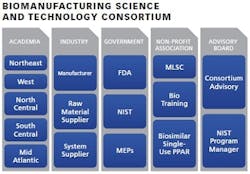
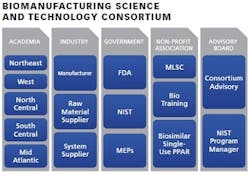
Launched in 2013, National Institute of Standards and Technology’s (NIST) Advanced Manufacturing Technology Consortia (AMTech) program is a competitive grants program intended to establish new or strengthen existing industry-driven consortia that address high-priority research challenges impeding the growth of advanced manufacturing in the United States.
The development of the biomanufacturing consortium will foster and support the fast-growing biopharma industry and the sustained leadership of the U.S. in that area. It is imperative to have a consortium of leaders to spearhead a “roadmap” for next-generation technology that will help to ensure future growth and stability of the industry. Regional meetings will be organized by the consortium in order to bring together leading experts to:
• Discuss the challenges and driving factors in upstream and downstream biopharmaceutical processing
• Identify the major technological, operational and regulatory barriers in biopharmaceuticals
• Develop a technology roadmap for biomanufacturing industry’s next generation
• Define a pre-collaborative workspace for companies, academia and regulatory authorities
Through a workshop-guided effort, the consortium aims to lay the foundation for a sustained partnership among the members that will foster U.S. competitiveness in the global market. Major participants include: Biogen, Genzyme, Shire, Genentech, Lonza, GSK, GE, EMD-Millipore, MLSC, Pfizer and Quincy College.
According to AMTech, “Without a roadmap, the industry’s current challenges cannot be transformed, innovative biomanufacturing systems cannot be pioneered, new technological standards cannot be set, and sustained global leadership of the U.S. biomanufacturing industry cannot be guaranteed.”
About the Author
Karen P. Langhauser
Chief Content Director, Pharma Manufacturing
Karen currently serves as Pharma Manufacturing's chief content director.
Now having dedicated her entire career to b2b journalism, Karen got her start writing for Food Manufacturing magazine. She made the decision to trade food for drugs in 2013, when she joined Putman Media as the digital content manager for Pharma Manufacturing, later taking the helm on the brand in 2016.
As an award-winning journalist with 20+ years experience writing in the manufacturing space, Karen passionately believes that b2b content does not have to suck. As the content director, her ongoing mission has been to keep Pharma Manufacturing's editorial look, tone and content fresh and accessible.
Karen graduated with honors from Bucknell University, where she majored in English and played Division 1 softball for the Bison. Happily living in NJ's famed Asbury Park, Karen is a retired Garden State Rollergirl, known to the roller derby community as the 'Predator-in-Chief.'