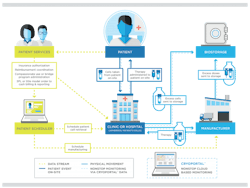
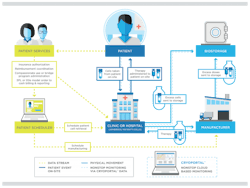
The increasing use of CAR T-cell therapy, immunotherapy and other autologous cellular therapies necessitate an integrated approach to the manufacture, shipment and storage of these new biologic products. The need for integration is largely due to the complexity of the end-to-end process for these therapeutics, which involves obtaining cell materials from a patient, sending the materials to the manufacturing facility, and then shipping the finished product back for administration to the patient at the dispensing clinic or hospital.
The process leaves no room for error in tracking or traceability, and the materials must be shipped and stored at the prescribed temperature range, whether it is 2–8 degrees C for the inbound apheresis material or at constant cryogenic temperature for the other legs of the supply chain. Moreover, the logistics of manufacturing and distribution must be closely coordinated with patient scheduling.
For those reasons, it is never too early for biopharmaceutical companies to initiate planning for clinical trials and commercialization of their autologous or allogeneic cellular therapies. As the accompanying chart illustrates, Figure 1, the integration process begins with scheduling the patient appointment at the clinic or hospital, an event that triggers several actions that must be precisely timed and sequenced. Some of these actions may need to be handled with a third-party hub services organization to coordinate patients and product access; handle clinical logistics; manage kitting, compassionate use or bridge programs, benefit identification, insurance authorization, billing and reimbursement, adverse events and complaint reporting; and provide medical and pharmacovigilance support.
To create an integrated clinical supply chain for complete end-to-end tracking, it is necessary to take the time to develop application programming interfaces to connect the disparate systems and information provided by the stakeholders involved. This will ensure tight coordination and transparency of all elements of the supply chain.
Figure 1. Sample schematic for shipment of biologic materials for autologous therapies.
For the patient scheduler, the key outputs are scheduling the cell retrieval from the patient and scheduling the product manufacture. Depending on the integration scheme, the latter may involve scheduling of shipments from the manufacturer to the clinic/hospital where the therapy is administered to the patient.
Each step in the process involves transmission of a data stream to facilitate the precise sequencing and timing of shipments, as well as continuous monitoring, recording and documentation of the condition of the biologic material (and its packaging) at all stages of the journey. Any variation in temperature or packaging orientation, or any interruption in the supply chain, can delay delivery and administration of the therapy to the patient and may also impact clinical trial results by compromising data integrity as well as product efficacy and safety.
The complexities surrounding the scheduling of patient events and the manufacture and distribution of cellular therapies have spurred the development of logistics systems for managing shipments, monitoring the condition and location of materials, and providing purpose-built packaging. These systems incorporate integrative technologies that enable a continuous flow of material and data to optimize the supply chain, ensure temperature control and lend visibility to shipment tracking, condition monitoring and storage management typically using dedicated liquid nitrogen cryogenic freezers with comprehensive inventory management capabilities. When fully integrated with clinical software, these technologies can be particularly useful to patient schedulers by creating shipping orders that coordinate smoothly with patient site visits.
Cold chain distribution requires extensive pre-planning of the supply chain and digital integrations for clinical trials and commercialization. For biopharmaceutical companies, it helps to partner with a provider of advanced, custom-built logistics management technologies.
Providers offering cloud-based platforms can simplify and provide transparency to the entire cold chain process by condensing ordering, tracking, paperwork, customs documentation, reporting and communications into a single integrated data stream that also consolidates all logistics, condition monitoring and equipment qualification data for every shipment. Such platforms can provide streamlined communication between all parties (i.e., sponsor, manufacturer, shipper, hospital/clinic, and patient scheduler), preloaded documents and supplementary information, carrier and shipping lane optimization, 24/7 access to shipment location and condition, and proactive customizable alerts. These systems can thus help optimize workflows and shipping lane viability.
Logistics management platforms work best when integrated with condition monitoring systems that provide real-time information about a company’s shipment, as condition management is essential to ensure the integrity of fragile, temperature-sensitive biologic materials. These systems provide visibility of both the conditions and location of a shipment at each leg of the supply chain, generating data to document shipment integrity during transit. Advanced monitoring systems can track and create specific histories of key shipment attributes that may impact the quality and/or timing of delivery including location (tracked via GPS, cellular and Wi-Fi triangulation), barometric pressure, orientation (i.e., the container’s physical position), time and location of shock events, internal and external temperature, light (as an indicator of security breaches, compromised packaging or premature openings), and humidity/moisture level. This data is collected via wireless communication and transmitted and reported at a customized frequency.
Logistics management providers can ensure compliance with regulations for distribution and storage of biologic materials, such as those issued by the International Air Transport Association (IATA). Purpose-built packaging such as liquid nitrogen dry vapor shippers fully comply with IATA standards and are classified as non-hazardous by the IATA, requiring neither special labeling nor handling. A logistics management partner can facilitate preparation of documents and permits — particularly to expedite any customs documentation and clearance — to ensure safe and timely delivery, while also streamlining documentation by automatically generating carrier-specific air bills and shipping labels.
When biostorage is also required, it is important to ensure that the logistics partner chosen also complies fully with Good Distribution Practice (GDP) standards, which require provision of validated cold storage equipment and freezers that are temperature-mapped and connected to backup generators, as well as temperature monitoring and alarms, to ensure an uninterruptable power supply and round-the-clock environmental control. Additionally, the logistics management partner should deploy a validation team to document that all equipment meets or exceeds US FDA requirements and guidance documents as well as quality standards issued by other regulatory bodies.
By necessity, logistics management providers focus heavily on quality systems, an area that includes auditing of records pertaining to temperature, access and inventory control, as well as documentation of training programs, including cross-training of employees to ensure adequate training in security, continuity, fulfillment and inventory policies and procedures. Quality systems also encompass safety, and a logistics management partner can help a biopharmaceutical company document implementation of measures to ensure biological, chemical, electrical, fire, physical and radiological safety, as well as of safety measures for the types of materials (e.g., liquid nitrogen, dry ice, carbon dioxide) that are shipped and stored.
Partnering with a logistics management provider can help ensure the seamless transit of biological materials from patient to manufacturer and back to the patient. The integrative technologies that facilitate timely delivery of autologous or allogeneic cellular therapies under constant and precise temperature control can enable administration of therapy to the patient at the appropriate time and place, whether as part of a clinical trial, a compassionate use program or provision of a commercialized product. While the benefits of early planning for cold chain shipping are considerable, biopharmaceutical companies may lack the requisite capabilities and expertise to implement such plans. That is where integrated logistical management service providers with cloud-based management platforms, condition monitoring systems and customized packaging can help.