Continuous Low Moisture Wet Mass Granulation: A Demonstration of Potential
| Note: To watch a video of the processes described in this article, click here. For a previous article by this author on the potential of continuous processes in pharma, read here. |
Wet mass granulation is the basis of most tablet formulations in the pharmaceutical industry. It is, at present, a time consuming and multi-stage process that involves considerable time, energy consumption, operational steps, and machinery. Much of the effort and time involved is based on the assumptions that are used in how the process must be conducted, as opposed to how it can be simplified and the operation simplified. Developing an alternative process that would simplify and streamline the process would offer significant improvements for the pharmaceutical industry.
The benefits of continuous processing are well known (References). FDA draft guidance encourages innovation in manufacturing technology, including continuous processing. The basic principle of Quality by Design (QbD) is that “the quality cannot be tested into a product, but must be built in by design”. Specifically, the draft guidance cites: “Facilitating continuous processing to improve efficiency and manage variability”.
Although the perceived benefits and advantages of Continuous Processing are known, the lack of applications in the pharmaceutical industry and the historical emphasis on batch type processing, has meant reluctance to view continuous processing as a legitimate option to consider. One method to clarify the possible role of continuous processing in the Pharmaceutical Industry would be to document a continuous process production demonstration of a widely used process.
Wet mass granulation is one process that is widespread in pharmaceuticals that would be understood in concept but also would provide an objective way to show comparative methods between batch preparation and continuous processing.
Background of Wet Mass Granulation:
About 70% of the pharmaceutical products are solid dosage forms (tablets, capsules, oral powders and powders for solutions or suspensions). The efficiency of tableting, encapsulation and powder filling processes depends on the quality of the granulation which can be processed on high speed machines. Adequate performance requires a granulation process that insures good quality granulation suitable for these processes. Beside direct compression, the granulation processes of choice have been: wet granulation or roller compaction. Both of these granulation processes utilize multiple unit operations, requiring additional pieces of equipment for each unit operation. In addition, many granulating unit operations require critical control of variables. Recently, the use of Process Analytical Technology (PAT) has made it possible to better control these variables, but it has not been very easy or simple or inexpensive. Inherently, because they involve multiple steps done in sequence, these processes do not easily lend themselves to the possibility of continuous processing.
Objective
Performing wet mass granulation on a continuous basis would simplify the process, reduce the time involved, reduce human error, batch deviation or loss, reduce the number of steps and machinery involved, and significantly reduce the cost to perform this operation. At the same time, it is necessary to achieve equal to or better than product requirements that are necessary for volume production of tablets and the tablet press concept. This paper will present the findings of a demonstration conducted on a wet mass granulation process that will show the ability to achieve the desired objectives.
Background
The most common steps involved in the wet granulation process are:
1. Preparation of pre-blend.
2. Addition of granulating fluid or solution/suspension
3. Mixing or kneading to create wet mass or wet granules
4. Drying of wet granules
5. Sizing of dry granules
6. Mixing of other excipients
7. Mixing of lubricant
It is possible to carry out several of these steps in a high shear or a planetary mixer followed by tray or fluid bed drying. Another alternative that will combine several steps is to use a fluid bed granulator, but a granule sizing operation is still needed as well as the mixing operations for excipients and the lubricant. Most of the efforts directed towards continuous wet granulation have been directed towards improvements on the equipment side of the equation in order to overcome the challenges involved in traditional wet granulation. However, little or no innovation has been directed towards removing the road blocks in the wet granulation process that impede the conversion to continuous processing.
Defining the Existing Process
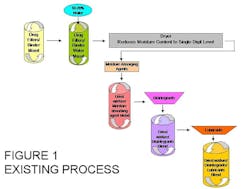
The process involves mixing dry powders, consisting of excipients, and active pharmaceutical ingredients, with a liquid binder causing the wet mass to agglomerate into granules. Drug fillers and/or binders are first mixed to make sure of mixture homogeneity. At this point, the resulting granulated mixture is combined with water in the range of 10-20%, and again mixed to insure homogeneity. The next step would be to dry the resulting mixture to reduce the moisture content down to the single digit level. The next step would be to add moisture absorbing agents and blending those into the granulated mixture. Next would be to add disintegrants, followed by a blending operation and a final addition of lubricants, and an additional combining operation. This process, commonly used, is the basis for most pill and/or tablet formulations. That process, utilizes a number of manufacturing processes and steps along with a number of different types of machinery that are involved in that process. In addition to labor, capital cost, and time of manufacture, there are also significant energy costs, particularly in the drying cycle, for the product.
Figure 1 outlines the existing process.While not all tablet formulations may follow this exact sequence and some may have significantly different percentages of the different ingredients. They may have different ingredients or may have single or multiple ingredient additions in the process. However, the wet granulation process follows this general type of sequence.
Drying and sizing are the major road blocks in the path of development of a truly continuous wet granulation process. In addition, a significant PAT investment is needed to control the granulation end point, moisture removal, and sizing of such granules. It would be possible to convert wet granulation to a continuous process, if it was possible to eliminate drying and sizing steps from wet granulation as well as make the granulation end point less sensitive to shear and residence time.
The Continuous Low Moisture Wet Mass Granulation addresses these areas and opens up the possibility of significant change in the process. In this process, only the necessary amount of water needed to create granules is added, but not any excess water which would need to be removed by drying; therefore no drying step needed. The amount of water used in the process is usually 1-4%, of the finished product and thus the agglomeration process is not very sensitive to shear and residence time. This process creates only small, relatively spherical granules that do not need sizing. As a result, this process only has two unit operations, agglomeration and mixing
Overview of Demonstration
The Continuous Processor manufactured by Readco Kurimoto was utilized as the mixing equipment for performing all these operations described above. The powders were introduced as dry ingredients and metered through gravimetric screw feeders mounted on precision loss-in-weight scales for closed-loop verification. The liquids were metered through Peristaltic pumps using downstream coriolis flowmeters for closed-loop verification. The entire blending process was controlled through a central touchscreen operator interface containing all operation, recipe and tuning parameters for both the Continuous Processor and ancillary metering equipment.
The continuous mixing machine is a twin-shaft co-rotating device which contains individual mixing elements that intermesh. These mixing elements can be interchanged to make mixing concentrations and/or characteristics to be tailored to specific situations and to affect particular chemical reactions. By trading time for intensity, all of the combining operations can be done in a very small space, and in a very short time frame. This process also reduces the discharge of costly energy sources to the atmosphere.
The machine used for the test has a mixing barrel approximately 35-inches long and is powered by a 5-HP motor, capable of running at speeds from 25 to 300 RPM.
Test Setup
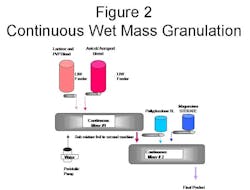
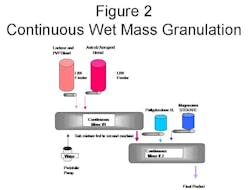
Two continuous mixing machines of a small production size were setup in sequence, with the output of the first machine fed directly into the input port of the second machine. The first mixing machine was fed a small percentage of water (1-4% by weight), and the binder which mixed with a blend of lactose and PVP12. The moisture absorbing material, a blend of Aperoperl 300 and Avicel LM200 was also fed into the mid point entry of the first machine. The resulting mixture was generated at the throughput rate of 80#/hr. with a total cycle time of less than one minute. That resultant was then fed directly into the inlet of the continuous mixing machine #2 where the disintegrant (Polyplasdone XL) was added. The final ingredient is a lubricant (Magnesium Sterate blend), and this ingredient was metered into the mid-port feed port of the second continuous machine.
Figure 2 is a schematic of the production set up.
The feeders and pumps were calibrated to specific formula levels and feed rates prior to testing. Graphs were generated from the data collected during calibration, and the feed rates quantified to match the intended levels. Total foot print area consumed by this production demonstration was 8’ by 8’ by 8’.
The blending control system was designed to integrate all discrete components onto a single Allen-Bradley ControlLogix PLC hardware platform for ease of use, maintainability, and reliable operation. All process temperatures are monitored directly by a PLC thermocouple card. All shaft speeds are monitored directly by a PLC tach encoder card. Both the powder and liquid loss-in-weight scales are monitored directly from load cell amplifiers by the PLC over an Ethernet IP network. All valves and motors are controlled directly for the PLC I/O cards.
The result of the test was a product throughput rate of 80#/hr. This production rate was achieved using the smallest size production machinery manufactured by the company. Larger scale production rates are available using larger capacity machinery. The total cycle time for this process from entry of the first ingredient to collection of the finished product was less than a minute and a half. The total horse power draw during operation was less than 2 HP. The resulting material was judged to be well mixed, uniform, and free flowing. A result of the table testing capability of the finished material indicated that it satisfied all requirements of the wet mass granulation properties for tablet compaction.
This type of machinery used and the results of this process have been reviewed in the literature, and this demonstration is confirmation of the use of the continuous process for this and other powder processes. The ability to process powders continuously, with low moisture content, and with multiple ingredient additions during the process, is a significant factor in process improvement in the powder processing industry.
Conclusions
There are several factors that can be concluded as a result of the success of this demonstration.
The continuous mixing machine is capable of “wetting” or homogenously mixing powders when the per centage of moisture addition is in the 1 to 4% range. This is accomplished be simply adding only the desired level of moisture directly to the mixing chamber during operation, and achieving a consistent, homogenous result.
The continuous mixer is doing the work of this process in a small, controlled chamber so that the work energy required is vastly reduced. Because the “wetting agent” is only introduced at the desired level for the finished product and there is no need for a drying step in processing, the Horse Power consumption for the entire process is less than 2 HP for 80 LBS/HR.
The use of gravimetric feeders along with the commensurate electronic control system, demonstrates that it is possible to attain significantly high standards for accuracy of feed rates. The advertised standard from major feeder suppliers is “1/4% gravimetric accuracy @ 2-sigma based on 60-second catches.” The process is totally scalable up to through put levels of 60,000 lbs/hr, by the use of larger machines. The use of existing technology in control systems has demonstrated the ability to control the results to meet industry standards for control.
It is possible to consider continuous wet mass granulation as an alternative to the existing method to produce tablet and pill formulations. This potential could allow manufacturers significant cost and productivity savings and streamline operational concerns.
References
Mollan, Matthew J. and Lodaya, Mayur, Continuous Processing in Pharmaceutical Manufacturing.
Crosby, Tim. Designing For the Future of Continuous Processing. Pharmaceutical Processing June 2009.