MES Systems Help Facilitate Modern Regulatory Compliance
Manufacturing Execution Systems (MES) enable pharmaceutical manufacturers to improve performance and reduce operational cost, while simultaneously increasing compliance with regulatory requirements.
The MES plays a critical role at the center of the manufacturing operation, connecting shop-floor personnel, equipment automation, logistics, sales and planning. Its ability to align these different functions and provide visibility to key stakeholders drives performance and compliance across the organization.
In order to facilitate regulatory compliance and data integrity, pharmaceutical manufacturers are eliminating the use of paper batch records in favor of electronic batch recording and MES. In doing so, they are recognizing the importance of integrating MES technology with Enterprise Resource Planning (ERP) and other complementary systems, to best ensure high manufacturing process quality and more effective, efficient regulatory compliance.
INDUSTRY CHALLENGES
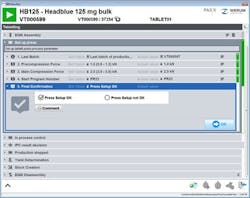
Operator at PAS-X EBR screen.
Currently, various healthcare trends around the world are combining to add new layers of complexity over an already complicated atmosphere for pharmaceutical manufacturers. Indeed, today’s drug producers face unprecedented challenges, including smaller batch sizes due to increased drug segmentation, expectations of shorter time-to-market, and increased regulatory complexity that includes current and pending serialization mandates. These are new challenges requiring innovative solutions.As a result, manufacturing IT solutions must respond to all these changes, and must combine software and services in a manner that addresses both current and future needs. The most important common denominator is this: MES solutions need to be comprehensive — they must be functionally complete, flexible, reliable and easy to operate.
They must also help ensure compliance, the most fundamental capability of a comprehensive system. MES technology can help the pharmaceutical and biopharmaceutical industries achieve compliance with regulatory authorities in various locations worldwide, including GMP requirements and FDA mandates for electronic records and e-signatures. Increasingly, compliance with anti-counterfeiting requirements is also coming into play.
PRODUCT VALIDATION
Let’s consider the role MES can play in the validation of computerized systems. The fundamental prerequisite that qualifies any system for validation is that clearly defined quality assurance (QA) policies have been observed throughout all project phases.
In essence, implementation steps must be concluded with verification proving that all regulatory requirements for that step have been fulfilled. An ideal MES software product will meet all of these requirements. Its functionality will comply with regulatory requirements, such as the ones set forth in the FDA guidelines 21 CFR Part 11 and 21 CFR Part 210/211, as well as EU GMP guidelines including the GMP Annex 11 for Computerized Systems Guidelines.
THE MANUFACTURING BACKBONE
PAS-X dialog for Electronic Batch Recording (EBR).
It’s useful to think of MES as the “manufacturing backbone” of the pharma industry — as technology that covers all needed functionalities while remaining focused on the industry’s specific requirements. MES software serves to integrate different business functions like process development, commercial production, and packaging in one central platform, integrating not only a company’s ERP system with the production equipment on the shop floor level, but also with its complementary systems, such as QMS, LIMS and DMS.An ideal MES solution provides a great number of benefits, such as improved production process efficiency (e.g. “review by exception” to speed up the release of batches), reduced time to market, optimized and secured process and product quality (e.g. fast, reliable and secure data entries and improved process security due to early detection of deviations), and alignment with compliance requirements.
RAPIDLY CHANGING REGULATORY COMPLIANCE REQUIREMENTS
Because pharma and biotech compliance requirements evolve at a rapid pace, MES solutions must adapt to these changes. Werum IT Solutions remains responsive by continually tweaking and making improvements to its PAS-X MES solution, which is run by a wide range of global and regional or mid-sized pharmaceutical and biotech companies. PAS-X 3.1.8, the latest version, both facilitates regulatory compliance and ensures high quality manufacturing processes.
PAS-X KPI Solution: operator dashboard – all important performance data at a glance.
Werum finds collaboration with pharmaceutical manufacturers themselves to be a crucial component in defining the future strategy for its MES solution. This May, Werum hosted its annual PAS-X User Group Meeting in the U.S., which brought together pharmaceutical vendors and manufacturers. The events provide a forum for MES users to discuss industry trends, present their best-practice MES implementations and exchange experiences with peers. The company listens carefully, and responds to the identified needs and trends by creating more useful and responsive solutions. The next PAS-X User Group Meeting will take place at Werum’s headquarters in Lüneburg, Germany, on December 1 and 2.Thanks to greater availability of sophisticated MES solutions today, pharma and biotech manufacturers are able to achieve compliance, quality and performance with greater success than ever before. Companies are reducing their costs in terms of waste, rework and defects, and increasing their productivity through real-time data analysis that drives process improvements.
ABOUT THE AUTHOR
Lars Hornung is senior director global sales at Werum IT Solutions (www.werum.com), a supplier of manufacturing execution systems (MES) and manufacturing IT solutions for the pharmaceutical and biopharmaceutical industries. Werum is headquartered in Lüneburg, Germany, and has many locations in America, Europe and Asia. Werum is part of Medipak Systems, the Business Area Pharma Systems of the international technology group Körber.