Solvent removal is a key stage in many pharmaceutical processes. It commonly occurs through a drying process, in which the solvent evaporates from the product in a process vessel such as a vacuum, tray or rotary drier. Historically, labs confirmed successful solvent removal by testing a sample at the end of the process. The test, known as loss on drying (LOD), measured the amount of residual solvent in the sample, and a failing result meant that the lab must repeat the drying process, a costly and disruptive rework.
Solvent removal is a rate-limiting step in many pharmaceutical manufacturing processes. This means that drying errors can have consequences that affect the efficiency of the entire operation.
PAT DEVELOPMENT
To make the solvent drying process more effective and efficient, many companies have turned to process analyzers — analytical instruments that take regular readings of solvent levels while the process is occurring, rather than only at the end. In 2004, the U.S. Food and Drug Administration (FDA) validated this approach when it launched its process analytical technology (PAT) initiative.
Many techniques are used for analysis during the drying process. One, near-infrared (NIR) spectroscopy, is unique because it uses probes to sample drying materials directly. Making physical contact with the active pharmaceutical ingredient (API) in mid-process has several drawbacks, however. First, driers with moving parts (such as paddle driers) usually must be temporarily stopped to allow the sampling probes to safely reach the sample. Probes can also become coated with the API over time, causing them to lose accuracy. Finally, NIR sampling probes only measure the dryness of the small percentage of the total sample they physically touch; though data from several probes can be aggregated to give a reasonable estimate of the total residual solvent in a sample, such an estimate can never be entirely accurate.
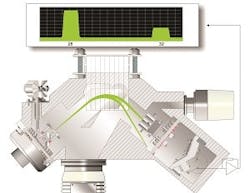
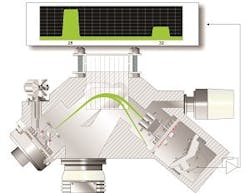
Another analysis technique used for solvent drying processes is gas analysis mass spectrometry (MS), which offers several benefits as compared to NIR spectroscopy. Instead of using sampling probes to measure the residual solvent in the API, MS systems sample the headspace above the drying material and measure the concentration of solvent evaporation byproducts. This is a significant improvement over NIR for several reasons. First, MS analysis is non-contact and, therefore, it does not require any interruptions in the drying process for sampling. Second, MS systems are not affected by heterogeneity in the drying product because they sample gases escaping from the API (and not the API itself, as NIR systems do). Lastly, because process mass spectrometers use non-contact sampling, it allows them to analyze gas continuously, which lets laboratory technicians save time and money by identifying the exact moment when the desired solvent concentration has been reached.
Gas analysis mass spectrometers are a significant improvement over NIR solutions, but they are prone to problems of their own – particularly early models based on quadrupole analyzers. These instruments were notoriously susceptible to contamination by headspace gases, especially gaseous hydrocarbons. Over time, this contamination causes analyzer drift that eventually requires the instrument to be taken offline for cleaning.
The problems with quadrupole-based analyzers have largely been addressed by magnetic sector mass spectrometry. Magnetic sector MS offers several advantages over quadrupole MS, including resistance to contamination, longer intervals between calibrations and improved precision and accuracy. Depending on the complexity of the gas mixture being analyzed, magnetic sector MS offers analytical precision between two and 10 times better than that of a quadrupole analyzer.
Beyond the inherent value of the technology, several design improvements can increase the performance of a magnetic sector MS system even further. Systems with laminated magnets, for example, can scan at speeds equal to those of quadrupole analyzers, offering both rapid analysis and the low maintenance previously mentioned. Enclosed ion sources can also improve magnetic sector MS performance by increasing sensitivity, minimizing background interference and maximizing contamination resistance.
VACUUM DRYING SAMPLING
The fact that magnetic sector MS analyzers operate at high vacuum makes them ideal for monitoring vacuum drying processes, but this is only true if technicians follow procedures correctly. It is vitally important that the pressure in the MS remains constant as the process pressure falls down to the vacuum levels required to dry the product. Poor control can cause the MS signals to rise and fall with sample pressure, which makes the collected data inaccurate.
Early magnetic sector MS vacuum drying systems struggled with this, especially when the vacuum process was complex. Many used only a single control valve — typically a voltage sensitive orifice (VSO) valve linked to a gauge monitoring the sample pressure. This worked well for simple processes: the VSO valve would open and close in response to sample pressure changes, keeping the pressure within the MS constant. Unfortunately, this technique suffered valve malfunctions and loss of control at pressures below 10 millibars.
This problem can be easily solved with the addition of a second control valve that works in opposition to the first — as one closes, the other opens. Together in a variable pressure inlet, a dual valve system allows an MS analyzer to handle sample pressures as low as 0.3 mbar.
Process instruments, no matter how capable and well-designed, are only effective when they are installed and calibrated correctly. Meeting the demanding standards of the pharmaceutical industry requires particularly rigorous testing. For MS systems, this testing is typically carried out at the production facility by connecting the new MS to the pharmaceutical company’s vacuum dryers. Testing on the production line obviously has a negative effect on throughput, which in turn can cause the testing process to be rushed. The end result includes inadequate testing, post-installation problems and damaged products. Understandably, going through this process has caused many pharmaceutical companies to lose confidence in MS systems.
To solve this problem, many producers of process magnetic sector MS analyzers are applying a widely used concept in the pharmaceutical industry: quality by design (QbD). Instead of testing the quality and capabilities of an MS system post-installation, as is traditional, producers ensure quality pre-installation by improving analyzer designs and performing more rigorous offsite testing.
Testing designs in-house — rather than on customer product lines — allows manufacturers to test MS designs over a much wider range of sample pressures, solvent combinations and solvent concentrations. Difficult challenges that push the limits of a magnetic sector MS — for example, switching between a dryer at the start of its drying run with high pressure and solvent concentrations and another reaching the end of its drying run, with opposite conditions — are much easier to test at a solvent drying test facility than on an actual pharmaceutical production line.
Offsite testing also allows engineers to change the design of an instrument, if necessary, based on observations from testing. Using a practical process improvement (PPI) approach, engineers can use test facility data to further ensure that quality is designed into the instrument. Most important, a combined QbD/PPI methodology helps ensure that finished magnetic sector MS analyzers can be installed in production lines with minimal process interruption.
Because they are rate-limiting, errors in solvent drying processes can have significant upstream and downstream repercussions for pharma production processes. Magnetic sector MS improves efficiency of the solvent drying process because it does not interrupt the drying process and is not affected by heterogeneity in the drying sample, as is true of LOD and NIR testing. Magnetic sector MS instruments are also several times more precise than quadrupole analyzers and require significantly less maintenance.