Improving the Efficiency of Your Production Facility
Despite the cautious optimism following the recent economic crisis, people who design, manage and maintain production facilities are still facing tough decisions. Competition from emerging economies such as the BRIC countries (Brazil, Russia, India and China), slow consumer response, rising prices for raw materials and uncertainty about the immediate economic future, are just a few of the issues that are continuing to challenge production managers today.
In this context, what can managers do to ensure the continued viability of their operations? A certain number of companies have gone for off-shoring and outsourcing to low-wage countries. But with production and quality issues becoming more and more of a concern, this option is beginning to lose some of its shine, and some managers are hesitant to take this route as it can complicate an already complex and over stretched supply chain. Another popular option is cost cutting, but it would appear that most of the “low hanging fruit” has already been picked, with the danger now being that companies will be trimming muscle instead of fat.
For pharmaceutical firms the picture might be a little more complicated in that, for many, the primary concern is the development of new products, which soaks up huge amounts of research and development. The actual production is either the last step in an intricate series of events or outsourced altogether. An already complicated process would have an added dimension of complexity in that one is often dealing with heightened safety and quality standards, far greater than those found in most other industries.
Numerous pharmaceutical manufacturing procedures involve large amounts of some raw material passing through a series of processes. Invariably done as a batch operation, this activity could also be thought of in terms of a flowline. The material flows in one direction. There are a series of precedent constraints. At each step some “work” or transformation takes place until the material in process has passed through all of the stages emerging as a finished product at the last step. An example would be the steps involved in changing some active pharmaceutical ingredient (API) into a tablet.
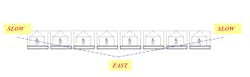
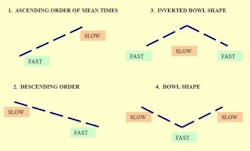
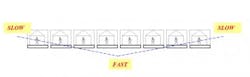
When dealing with the production of pharmaceuticals, quite often the focus is on scheduling and capacity planning, with the goal being to get the most output out of a given facility, given a limited amount of resources. But our research has shown that there might be another way to increase output, reduce idle time or lower the average amount of material in a given linked set of processes. From Starving to BlockingThe aim of this article is to highlight possible ways for improving the performance of facilities having unpaced production lines, where operators are allowed to work at their own speed without the aid of an automated moving belt. We also take into account that these lines will necessarily suffer breakdown at times. Such lines usually have some storage spaces allotted in between the individual stations, referred to as buffers, for storing work-in-progress (WIP) units. This permits smoother production by avoiding stoppage of work due to an operator not having a work piece available (known as “starving”), or not being able to pass an item on to the next station due to lack of space (termed “blocking”). The importance of buffers should not be underestimated since they allow workers to process items relatively independent of each other. This type of production is called “asynchronous.”There has been a considerable amount of attention paid to achieving a “balanced” line, where every workstation along the line completes its task at equal rates, and passes the unfinished piece on to the next station. Some firms have made substantial investments in time, money and effort to bring balance into their lines, but is a balanced line desirable, or even possible? If we want to be realistic and flexible in our line designs, it needs to be recognized that different people work at different speeds, so perfect “balance” is a nigh on impossible dream. There are several reasons for this. One is the type of work performed—sometimes, because of pre-existing technological or precedence restrictions, it might be impossible to break down an individual job into a number of simpler tasks where each one has the same average completion time. We can imagine, say, a process where a series of chemical reactions are followed by centrifuging, drying, crystallization, and so on until the final packaged product is manufactured. We can’t simply run one of the reactions for half the necessary time in order to maintain a balanced line.The Human ElementAnother reason in these manual lines is human nature. Different people have different levels of training, education, skill, ability and motivation. For these reasons, the amount of time it takes for different operators to complete the same task will never be equal. Contrary to popular opinion, this is not necessarily a bad thing. Studies have shown that an unbalanced line where people work at different speeds can actually outperform a perfectly balanced line. By deliberately having at least one station slower than the rest (a bottleneck), higher production rates than those attainable by a corresponding balanced line were possible. An operations management approach known as the “Theory of Constraints” views bottlenecks as a normal everyday occurrence. By identifying the constraining bottleneck station and providing it with additional resources, better on-time delivery and overall performance are possible.Worldwide, billions of dollars are spent yearly on the design, running and maintenance of production lines. Even the slightest improvements in efficiency and operating costs can result in sizeable savings when considered over the anticipated useful life of a facility. So how can we design our unpaced lines to take best advantage of the natural working rhythms of our employees in so-called “unreliable” lines that can break down at any time?Simulated Case StudyWe conducted a computer simulation investigation into production lines having 5 and 8 stations. We incorporated station failure so as to duplicate real-life operating conditions as closely as possible. We assumed that individual workers fell into one of three categories according to their mean (average) service time; that is to say an operator could be fast, medium or slow. Four configurations of employee arrangement were looked at (Figure 1):- the fastest worker is located at the front of the line, followed by slower and slower operators—an ascending order (/).
- the fastest worker is placed at the end of the line, preceded by progressively slower workers—a descending order (\).
- the slowest operator is positioned in the middle, with workers getting faster as we go towards both the front and back of the line—an inverted bowl shape (/\).
- the fastest worker is stationed in the middle, with operators getting progressively slower as one moves outward in both directions—a bowl shape (\/).
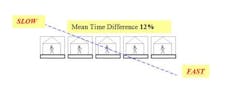
Another factor that we considered was the percentage degree of imbalance, i.e. the difference in speed between any two successive operators. Three degrees of line imbalance were viewed; 2% (slight), 5% (medium), and 12% (high). In addition, inter-station buffer capacities were set at 1, 2, 3 and 6 units.
Our objective was to find out which pattern (if any) of worker allocation would provide the greatest enhancement in performance by way of: 1) an increase in the throughput (output) rate; 2) a reduction in the overall amount of worker idle time; and 3) a decrease in the average buffer level.
Some of our results were surprising and might run contrary to the prevailing wisdom concerning production line management (Figure 2). We found that the best configuration resulting in increased throughput rates and lower average worker idle times was a bowl-shaped arrangement (fastest worker placed in the middle).
About the Authors
Tom McNamara and Sarah Hudson are professors at the Rennes School of Business, Rennes, France, in the Department of Finance and Operations. They can be reached at the following email addresses, respectively: [email protected] and [email protected]. Sabry Shaaban works within the Department of Economics Strategy and Organization at Groupe ESC La Rochelle, Cerege, France, and can be reached via email at [email protected].