Systech International
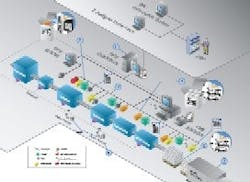
Systech's TIPS Serialized Product Tracking Solution manages all packaging line devices, including encoding and recording of the Electronic Product Code (EPC) serial numbers to the RFID tags and redundant bar codes.
The software verifies all data on the RFID tag and bar code, establishes the parent-child relationships in which the products are placed, such as cases and pallets, and stores the information in a secure database. The software then makes all the data available to the e-Pedigree, Authentication, Track and Trace, and other Business applications. The system handles all the real-time packaging level business processes including managing rejects, reworks and QA functions.
In addition, TIPS Serialized Product Tracking 7.0 solution has achieved SAP certification for integration with the SAP Auto-ID Infrastructure component of the SAP NetWeaver platform, which powers many SAP solutions for RFID. The software is one of the first fully 21 CFR Part 11-compliant solutions to earn this designation.
Currently piloted by several pharmaceutical manufacturers in the United States and Europe, TIPS Serialized Product Tracking supplies mass serialization of products at the item, case and pallet level to facilitate e-Pedigree, authentication, and track and trace. This serialization, using Electronic Product Codes (EPCs) on RFID enabled labels and bar code, is done on the packaging line and is integrated with existing packaging operations.
"For customers, this certification helps guarantee the seamless integration between our TIPS Serialized Product Tracking's comprehensive and reliable packaging line device management, data collection, and 21 CFR Part 11 compliance and the SAP Auto-ID Infrastructure," said Robert DeJean, SYSTECH's President and CEO. "This combination can help pharmaceutical manufacturers ensure product integrity across their supply chain."
Click here to access a 2-page, PDF-format datasheet on TIPS Serialized Product Tracking.