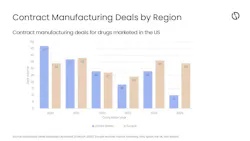
Contract manufacturing deals for drugs marketed in US shift to Europe: GlobalData
Despite the Trump administration’s imposition of tariffs on European Union (EU) pharmaceuticals, biopharma companies are increasingly moving to outsource manufacturing for the U.S. market to facilities in Europe, according to an analysis by data and analytics firm GlobalData.
Last year, the EU and U.S. announced a trade agreement that includes a 15% tariff on most EU exports, including brand name pharmaceuticals. However, GlobalData found that biopharma companies appear “undeterred” by the tariffs and are increasingly turning to contract manufacturing in Europe.
In 2025, U.S. contract manufacturing deals for FDA-approved drugs experienced their biggest decline in five years, according to the firm, while noting that the gap between U.S.- and Europe-based contract manufacturing deals was the widest yet with the EU securing more than triple the U.S. deal volume.
Last year, nine out of the 14 U.S.-based biopharma companies that outsourced manufacturing — including Johnson & Johnson and Vertex Pharmaceuticals — invested in a total of 13 Europe-based manufacturing deals, according to GlobalData. By comparison, the firm found less than half of these companies invested in U.S.-based facilities signing a total of eight contract manufacturing deals.
“The shift in manufacturing deals toward Europe highlights the limited impact that tariffs have had in steering biopharma companies away from European-based contract manufacturing,” Katia Djebbar, pharma analyst at GlobalData, said in a statement. “Such a shift may hinder the current U.S. administration’s plans to reshore domestic contract manufacturing.”
Djebbar called out the fact that Germany is “increasingly becoming an attractive and well-established hub for pharma manufacturing for the U.S. market.” She also noted that biopharma companies are selecting Europe for their in-house manufacturing facilities.
“Novo Nordisk and Eli Lilly announced investments of $501 million and $3 billion, respectively, to expand their in-house European manufacturing sites,” Djebbar said. “Following the recent FDA approval and success of Wegovy pill, which is currently the first and only oral glucagon-like peptide-1 (GLP1) receptor agonist available in the Western market, Novo is planning to expand its tablets facility in Ireland to meet the current and future U.S. market demand.”
With the launch of the Wegovy pill in the U.S., Novo Nordisk announced last month that it is adding manufacturing capacity to its Athlone, Ireland site to support current and future GLP-1 treatments. The Danish drugmaker is investing more than $500 million in the Irish facility.
FDA approvals fuel outsourcing penetration
Contract development and manufacturing organizations (CDMOs) are reaping the benefits of new drug approvals by the FDA, which bodes well for the pharmaceutical outsourcing and services sector, according to a recent report by William Blair analysts.
Their analysis found that 73% of FDA approvals that outsourced active pharmaceutical ingredient (API) manufacturing in 2025 were substantially above the 11-year average of 61% and in line with the 74% outsourcing rate recorded in 2024 — the highest level observed since 2015.
In the case of finished dose, the 65% of drugs outsourced last year represented the highest outsourcing rate the analysts have documented over an 11-year period — significantly exceeding the 50% average since 2015.
“Small and midsize innovators continue to outsource both API and finished dose production of their drugs far more often than large innovators, which when combined with our view that approvals will increasingly come from smaller innovators, appears to be a very favorable longer-term trend for CDMOs,” William Blair analysts said.
Related Listening
In this episode of Off Script, we spoke with Brian Doty, vice president of R&D and programs at the API Innovation Center (APIIC), about why API supply is often the cause of downstream drug shortages, the technical and economic limitations of traditional batch processing, and how continuous manufacturing can reduce process steps, improve control, and enable safer production.
About the Author
Greg Slabodkin
Editor in Chief
As Editor in Chief, Greg oversees all aspects of planning, managing and producing the content for Pharma Manufacturing’s website, digital products, and in-person events, as well as the daily operations of its editorial team.
For more than 20 years, Greg has covered the healthcare, life sciences, and medical device industries for several trade publications. He is the recipient of a Post-Newsweek Business Information Editorial Excellence Award for his news reporting and a Gold Award for Best Case Study from the American Society of Healthcare Publication Editors. In addition, Greg is a Healthcare Fellow from the Society for Advancing Business Editing and Writing.
When not covering the pharma manufacturing industry, he is an avid Buffalo Bills football and Buffalo Sabres hockey fan, likes to kayak, and plays guitar.