Digital transformation is best defined as the adoption of new manufacturing and business processes made possible by automation, communications and computing technologies represented by conceptual models such as data analytics, the Industrial Internet of Things (IIoT) and Industry 4.0. The pharmaceutical industry has seen great advances in transformative technology, and it’s becoming increasingly evident that many of these innovations can give manufacturers an edge.
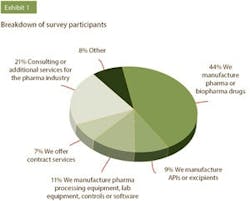
A recent Pharmaceutical Manufacturing survey separately asked over 200 drug manufacturers and equipment and services suppliers(*) their thoughts on the industry’s progress in terms of digital transformation. When asked which areas of pharma manufacturing they felt would see the most benefit from increased digitalization, both manufacturers and suppliers noted the same top three target areas: plant-floor production, quality control and supply chain management.
Close analysis of the survey results against the backdrop of today’s pharmaceutical industry landscape reveals that while the utopian picture of the benefits of digital transformation has been painted, in order to start realizing these benefits on the plant floor and throughout the supply chain, pharma needs a structured plan of action.
CHALLENGES LINGER
Several commonly discussed stumbling points still need to be dealt with as pharma continues along its digital path. When asked to rank concerns surrounding smart equipment in the plant, both drug manufacturers and equipment vendors prioritized regulatory hurdles and integration.
It should come as no surprise that a lack of regulatory buy-in or understanding of new processes, whether real or perceived, remains a top-ranking concern. When vendors were asked what they felt to be the number one issue holding back their customers’ digital progress, the leading answer (28 percent) was fear of regulatory backlash. Interestingly enough, this area that has traditionally caused the most hesitation in pharma’s technological progress may stand to benefit most from digital transformation, as the smart factory’s ability to automatically generate reliable audit trails, real-time reporting and electronic signatures promises to ease compliance burdens.
Integration concerns ran a close second among both survey groups. Respondents noted potential challenges surrounding the integration of new technology with existing lines or equipment. Despite the recent push towards automation, there are still many legacy systems in operation on the pharma plant floor that lack the capability needed to connect to high level automation systems or devices.
In fact, when vendors were asked if they felt that pharma companies were actively looking to replace outdated equipment with updated and improved robotics and automation, only 17 percent said that, overall, pharma manufacturers were actively updating. Just under 42 percent took a more neutral stance, noting about a 50/50 mix of companies actively updating.
TECHNOLOGY AS AN ENABLER
When ranking their concerns surrounding smart manufacturing plants, tech innovation was toward the bottom of the list according to pharma and dead-last according to vendors (a little tooting of their own horns there). But perhaps vendors are warranted in giving themselves a pat on the back, as it seems that overall, the industry is confident that the level of technology being offered is advanced enough to do the job.
These results are in consonance with the manufacturing sector in general, as well. At the recent Smart Industry 2017 conference - an annual event geared toward accelerating the ongoing digital transformation of manufacturing - Jose Rivera, CEO, Control System Integrators Association (CSIA), spoke about new opportunities and challenges for manufacturing’s automation ecosystem. Commenting across all manufacturing industries, Rivera pointed out that previously, “technology was more a barrier than an enabler.” The current technology revolution, however, says Rivera, “will allow manufacturers to get closer to their supply chains and plants.”
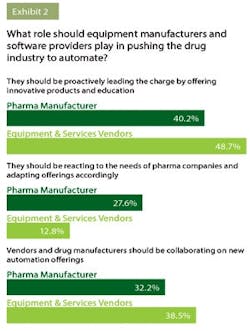
Much of the responsibility for advancing this technology in pharma has fallen on the shoulders of industry vendors. When asked what role equipment manufacturers and software providers should play in pushing the drug industry to automate, 43 percent of everyone surveyed felt that vendors should be proactively leading the charge. About 34 percent felt that there should be a collaboration between pharma and vendors (see exhibit 2).
MORE GOOD NEWS
It is clear that digital transformation is not an initiative that can be spearheaded by a single individual. But how does a traditionally conservative industry execute cultural buy-in? The top is not a bad place to start.
“We are working with several different pharma companies and we are very impressed with how digitally savvy the leadership is - there seems to be a good deal of awareness of what’s possible. There’s a curiosity at a leadership level, as they look around their companies and decide what they should do where and how to best approach transformation,” says Ryan Smith, Ph.D., VP of Engineering at Sight Machine (see Ryan’s article here). Sight Machine offers a state-of-the art analytics platform used by Global 500 companies to improve efficiency of manufacturing operations.
But while buy-in from the top is necessary, it’s not enough. “Organizational readiness is the key component to success. There has to be a minimal threshold of organizational readiness, and that does not just mean support at the top. It means support at all levels, especially on the plant floor,” continues Sudhir Arni, Sight Machine’s lead for Digital Manufacturing.
According to Pharmaceutical Manufacturing survey results, however, employees on all levels are noting a rising comfort level with the new computing, control and communications technologies represented by the IIoT. Over 81 percent of pharma respondents noted an increase in automation-related comfort in the industry, while 68 percent of vendors agreed that the pharma workforce is becoming more comfortable.
Encouragingly, when it comes to designing or upgrading its manufacturing facilities, the majority (64 percent) of pharma respondents and vendors (55 percent) feel that digitization is an important part of the discussion. Just under 28 percent of pharma respondents feel that digitization is leading the discussion, while 33 percent of vendors agree with that assessment.
EXECUTION VS. ASPIRATIONS
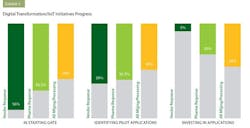
With the right tools available and a “big picture” view of IIoT benefits laid out before them, pharma’s next steps are crucial. Throughout the survey, pharma manufacturers were consistently more optimistic about digital transformation progress than their vendors. When asked to describe the pharmaceutical industry’s collective progress on digital transformation/IIot initiatives, 44 percent of pharma manufacturers felt the industry was at the starting gate, with a focus on learning and exploration. Just under 37 percent felt the industry was in the next stage, now identifying early applications to pilot. And 20 percent felt pharma was in the most advanced stage, having identified applications and made investments to match (see exhibit 3).
When vendors were asked the same question, however, the survey found 56 percent saying pharma was at the starting gate, and 39 percent claiming pharma was identifying early applications to pilot. Less than 5 percent of vendors noted actual investments being made.
The differing opinions of pharma manufacturers and vendors aside, the bulk of the industry appears to be stalled in the earlier stages of digital transformation. Here we see the true digital transformation crossroads: pharma understands the power of digital, but can’t continue its journey without investing in a tactical plan.
“What it comes down to is that it’s not really how you feel about it or how optimistic you are, but more so ‘what do you have in place to make it happen?’ Certain things need to be in place in order for an organization to be successful in digital transformation,” says Ed Jimenez, VP of Marketing at Sight Machine.
In order to be truly ready to execute versus taking a more aspirational approach, pharma needs a structured digital transformation strategy.
Again, we see vendors stepping up to the plate. Sight Machine, for example, has developed a Digital Readiness Index (DRI). Started as an internal tool to help customers assess where they stand, the DRI quickly morphed into a more formal methodology for evaluating a manufacturer’s readiness for digital transformation.
Manufacturers start by answering an online questionnaire and, based on the answers, the DRI uses a weighted scoring system to place organizations into one of five “Digital Readiness Zones.” For each zone, Sight Machine has recommended quick win projects and areas for investment to develop more advanced capabilities.
These “quick wins” are vitally important, as choosing initial applications that deliver value can stimulate company-wide buy-in and build much-needed momentum.
HOW PHARMA COMPARES
Perhaps the most important takeaway from the survey results is the message that pharma manufacturing is not yet - contrary to its reputation when it comes to initiating change - years behind other industries in terms of digital transformation.
Arni, who was the lead developer on Sight Machine’s DRI project and has conducted much of the company’s industry surveying, says the company is not seeing huge gaps in readiness from industry to industry. “In our experience, there is not much difference in terms of readiness between the pharma manufacturing industry and other manufacturing segments. The data from our Digital Readiness Index shows the pharma industry has the capabilities in place to move faster,” notes Arni.
In a recent Digital Transformation: 2017 State of Initiative report, Smart Industry surveyed professionals from across manufacturing, processing and related industries. When asked about their company’s progress on digital transformation initiatives, almost 72 percent reported not yet reaching the more advanced “identifying applications and making investments” stage. Additionally, more than half of those same respondents said they do not have a formal digital transformation strategy with timelines in place.
WHAT THE FUTURE COULD BE
The pharmaceutical industry understands the value of digital transformation. Survey results indicated that more than 70 percent of pharma manufacturers believe that a more automation-enabled pharma industry will lead to improvements in efficiency, productivity and quality.
With about 68 percent of those surveyed estimating that the pharma industry is somewhere between 5-10 years away from truly reaping all the benefits of IIoT and the “smart factory,” pharma’s next steps will determine its future.
What Sight Machine seeks to communicate with its Digital Readiness Index is a message that should be heard by all of pharma: “No matter where you are, there are always steps you can take today. Don’t keep waiting — get started.”
* For the purposes of this survey, “vendors” are defined as companies who offer pharma processing equipment, lab equipment, controls or software, as well as consulting or related services. “Pharma manufacturers” are defined as those who manufacture pharma or biopharma drugs, make APIs or excipients or offer contract services.
About the Author
Karen P. Langhauser
Chief Content Director, Pharma Manufacturing
Karen currently serves as Pharma Manufacturing's chief content director.
Now having dedicated her entire career to b2b journalism, Karen got her start writing for Food Manufacturing magazine. She made the decision to trade food for drugs in 2013, when she joined Putman Media as the digital content manager for Pharma Manufacturing, later taking the helm on the brand in 2016.
As an award-winning journalist with 20+ years experience writing in the manufacturing space, Karen passionately believes that b2b content does not have to suck. As the content director, her ongoing mission has been to keep Pharma Manufacturing's editorial look, tone and content fresh and accessible.
Karen graduated with honors from Bucknell University, where she majored in English and played Division 1 softball for the Bison. Happily living in NJ's famed Asbury Park, Karen is a retired Garden State Rollergirl, known to the roller derby community as the 'Predator-in-Chief.'