Improved Methods for Catalyst Recovery in Biopharmaceutical Production
They allow for fast reactions which require fewer steps than stoichimetric reagents. They operate at lower temperatures, decreasing capital costs and providing a higher degree of safety. One example is the Ullman reaction, which uses copper to couple two aryl halides to form the biaryl.
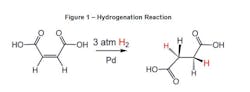
Other examples of coupling chemistries which use transitional metal catalysts are the Heck, Suzuki and Fukuyama reactions. In the transitional metals, the platinum group metals (PGM) are commonly used for pharmaceutical syntheses. PGMs include ruthenium (Ru), rhodium (Rh), palladium (Pd), osmium (Os), iridium (Ir) and platinum (Pt). Figure 1 shows an example of a hydrogenation reaction catalyzed by palladium.Catalyst Recovery
Catalyzed reactions can be broadly characterized into two categories: heterogeneous and homogeneous. In heterogeneous reactions, the phase of catalyst is different from that of reactants, i.e. solid, liquid or gas. However, it can also mean that the miscibility of the catalyst is different from the reaction mixture too. With homogeneous reactions, the catalyst and other reactants are the same phase and are miscible with one another. The type of reaction will have implications for the recovery of the catalyst post-reaction.
Solvent extraction, nanofiltration, chemical precipitation, and adsorption are catalyst recovery techniques typically employed in industrial and pharmaceutical processes. Nanofiltration recovery is used only with homogeneous processes and catalysts are immediately re-used in the liquid phase. Therefore, nanofiltration is not particularly amenable to incineration and recovery of the catalysts for later use. Solvent extraction and chemical precipitation tend to be somewhat non-specific and can lead to yield losses of the API.
Adsorptive recovery techniques can include materials such as activated carbon and ion exchange resins to recover either homogeneous catalysts, or homogeneous ligand and/or leachate. With a homogeneous catalyst, the carbon or resin will selectively absorb the catalyst. The catalyst is then stripped from the adsorbent for reuse or sent to a recovery specialist for incineration and recovery. For a homogeneous ligand and/or leachate, a fragment of a heterogeneous catalyst is shed and then dissolved in the phase where the chemical transformation is taking place. The catalyst is filtered from the batch and the ligand and and/or leachate that remains in solution is removed via resin or carbon treatment.
Whichever is the case, the spent heterogeneous catalyst (or carbon used to scrub the batch of catalyst leachates, ligands, and/or impurities) is washed with fresh solvent. These solvents can vary widely from process to process in pharmaceutical production, but typical organic solvents used in synthesis are MTBE, dichloromethane, tetrahydrofuran (THF), 2-methyl THF, acetonitrile, toluene, acetone, methanol, ethanol, ethyl acetate, or water.
The Problem with Carbon
Historically, adsorption techniques for catalyst recovery have involved activated carbon. Carbon is well characterized, it is relatively inexpensive and it is relatively effective in removing spent catalysts from process streams. However, carbon suffers from several drawbacks. First, it can be difficult to handle and filter from the process stream. This is usually overcome by operating the removal process with pre-packed carbon cartridges. The second, and more difficult, drawback is the non-specific adsorptive nature of carbon. Carbon will remove the catalyst, but it will also bind a percentage of the API. This leads to yield losses and increased production costs. The selective removal of the catalyst would be a more attractive adsorption option.
A More Selective Approach: Ion Exchange Resins
Ion exchange resin technology has been used for over 60 years in the biopharmaceutical industry, primarily for purification of APIs. Molecules as diverse as antibiotics, oligonucleotides and monoclonal antibodies have been purified using ion exchange resins. Specialized ion exchange resins are also used as scavengers for chemical synthesis, removing byproducts and un-reacted components. These uses range in scale from laboratory bench-top operations, such as solid phase extraction (SPE) strategies with combinatorial libraries, to full-scale production. One example of a scavenger resin at production-scale in industrial processing is the use of iminodiacetic functionalized resin for metal removal. The waste effluent before discharge still contains some low levels of heavy metals, about 5 to 20 ppm. A polishing unit containing the chelating resin installed in a packed-column merry-go-round configuration can bring the total concentration of metals below 0.1 ppm even in the presence of high concentrations of sodium or calcium salts.
Polymeric resin-based catalyst removal strategies have several advantages, including:
- A more selective removal of the catalyst, leading to higher recoveries of API
- Providing solid waste for incineration or selective desorption of the catalyst
Resin Requirements
The polymeric resins used for catalyst recovery need to meet several criteria. First, they must have excellent solvent compatibility so that they remain intact during exposure to the process stream. Resins that degrade or produce high levels of leachables will not be suitable candidates for this application. Second, the API should ideally not have any secondary interactions such as hydrophobic binding with the resin backbone. Secondary interactions could theoretically cause a reduction in API yield if the API becomes bound on the resin backbone. Third, low-swelling resins are desired, especially if used in column operations. If there is appreciable swelling during operation, pressure drop across the resin bed can become high thus reducing process throughput. Finally, a support package should be available for the resins in order to provide the appropriate information for regulatory requirements.
Factors Affecting Performance
With spend catalyst in an organic solvent, such as DCM, ethanol, THF or methanol, the functionalized resin may be used directly on the solution. Batch experiments can be performed easily by adding 5g of resin per 100mL of spent catalyst solution and shaking overnight at room temperature. The solution can then be analyzed to determine how much catalyst has been bound by the resin. The results of these binding experiments will assist in determining the amount of resin required for the recovery process. The full-scale operation can be performed in either batch, i.e. stirred tank, or in column mode. Column operations will generally be more efficient.Thiouronium resin chemistry is particularly selective for platinum group metals. However, it is unstable at high pH and is therefore recommended for only acidic and neutral pH conditions. A description of recommended pH conditions for the various resin chemistries is shown in Table 2.Conclusions
Specialty ion exchange resins are an excellent alternative to activated carbon for use in catalyst recovery operations. These improved polymeric chemistries selectively bind divalent metals, including platinum group metals, and can effectively recover catalysts while not adversely affecting API recovery. These yield increases help to improve economics while minimizing the environmental impact of the process.
References
1. Alexandratos, S.D., “Ion-Exchange Resins: A Retrospective from Industrial and Engineering Chemistry Research”, Ind. Eng. Chem. Res. 48 (2009) 388-398.
2. Barbaro, P. and Liguori, F., “Ion Exchange Resins: Catalyst Recovery and Recycle”, Chem. Rev. 109 (2009) 515-529.
3. Deepatana, A., and Valix, M., “Recovery of nickel and cobalt from organic acid complexes: Adsorption mechanisms of metal-organic complexes onto aminophosphonate chelating resin”, J. Hazardous Materials B 137 (2006) 925-933.
4. Jacobsen, R.T., “Catalyst Recovery – Part 1: Recovering Precious Metals from Catalysts – The Basics”, Chemical Engineering Progress, Feb. 1, 2005.
5. Kunin, R., “Ion Exchange Resins” Robert E. Krieger Publishing Co. 1958.
6. Luthra, S.S., Yang, X., Freitas dos Santos, L.M., White, L.S., and Livingston, A.G., “Homogeneous phase transfer catalyst recovery and re-use solvent resistant membranes”, Journal of Membrane Science 201 (2002) 65-75.
7. Nair, D., Luthra, S.S., Scarpello, J.T., White, L.S., Freitas dos Santos, L.M., and Livingston, A.G., “Homogeneous catalyst separation and re-use through nanofiltration of organic solvents”, Desalination 147 (2002) 301-306.
8. Nilsson, U.J., “Solid-phase extraction for combinatorial libraries”, J. Chromatogr. A 885 (2000) 305-319.
9. Scarpello, J.T., Nair, D., Freitas do Santos, L.M., White, L.S., and Livingston, A.G., “The separation of homogeneous organometallic catalysts using solvent resistant nanofiltration”, Journal of Membrane Science 203 (2002) 71-85.
10. Thompson, L.A., “Recent applications of polymer-supported reagents and scavengers in combinatorial, parallel, or multistep synthesis”, Current Opinion in Chemical Biology 4 (2000) 324-337.
11. Yoo, J.S., “Metal recovery and rejuvenation of metal-loaded spent catalysts”, Catalysis Today 44 (1998) 27-46.