Leveraging Green Metrics for Route Selection and Process Optimization
The pharmaceutical industry faces huge challenges today, as yesterday’s blockbuster drugs come off patent, the pace of mergers and acquisitions accelerates, and companies adjust to the dramatic changes in science and technology that have taken place within the past 10 years.
Where the old business model focused on getting fair returns for the high costs and risks associated with R&D, pharmaceutical manufacturers today must be far more consumer focused. Guiding their efforts are a number of indicators, including the unit price of the active pharmaceutical ingredient (API). Process optimization allows manufacturers to improve API production yields and efficiencies, with positive effects on process and product development, technology transfer to first commercial manufacture and site-to-site technology transfers.
To ensure that the best route to an API is in place whenever a new product is launched, Pfizer prefers to “co-design” commercial manufacturing processes. Within this framework, R&D and manufacturing collaborate closely after potential routes have been identified, to ensure that the best process chemistries are developed so that the commercial process will minimize both manufacturing cost and environmental impact.
As its co-design strategy has evolved, Pfizer has refined its use of vital cost and environmental modeling platforms. As a result, scientists and manufacturing professionals now have access to the detailed data they need for final route selection and optimization. This article will take a brief look at how Pfizer is using modeling to improve process optimization and route selection to improve efficiency, reduce cost and environmental impact, and enhance collaboration throughout R&D and manufacturing.
Methods of Green Manufacturing Metrics and Cost of Goods Analysis
In the past, teams had to access important information from myriad sources, ranging from paper notebooks to individual queries, which limited access to critical data. This information would be distilled into very simple form, based on the consumption and cost of regulatory-compliant starting materials and the primary building blocks of the molecules considered for commercialization.
In addition, R&D and manufacturing each developed different cost models, using customized spreadsheets. On the research side, processing costs were often estimated without knowledge of actual cycle times, based on a cost-per-step per unit mass of product, with some graduation of that cost as a function of annual production volume. This method did not incorporate information about disposal costs or relative production of waste per unit mass of product. Its benefits were clearly seen during the earliest stages in the product lifecycle, for instance, when a choice had to be made between five or more potential routes with little lab or scale-up experience of any of the options.Though it generated results quickly through spreadsheet analysis, it could not provide sufficiently granular information on itemized costs or environmental metrics to add great value to route selection for long-term manufacture. Over time, detail was added to the cost and environmental models, to improve the depth of results and using primary data to drive commercial route selection. Unfortunately, this was often done via custom spreadsheets developed by each portfolio research project leader and shared with his or her manufacturing colleagues.
Meanwhile, the manufacturing organization was also generating its own version of the cost of manufacturing campaigns, with details that were manufacturing site specific but did not represent the long-term, optimized commercial process. Merging these two different estimates was often difficult, and information transfer between R&D and manufacturing less than optimal.
These approaches featured little if any measure of green manufacturing metrics, while the customized nature of the spreadsheet analyses made them difficult to apply.
Though highly detailed, these spreadsheets were adapted each time a new synthetic step or alternate route was analyzed. Reconfiguring them to fit particular scenarios meant that these platforms were prone to error. The unique structure of each, relative to individual project leaders, meant that the results were difficult for managers, who were considering the entire scope of portfolio development and commercialization, to adequately assess.
As a result, management introduced a standardized template structure upon the research and manufacturing spreadsheet approaches in an attempt to capture stepwise information and various route configurations. For instance, they improved the calculation of processing costs by using generic cycle time estimates for each unit operation of a synthetic step. Total cycle time was then multiplied by a generic cost of multiple-vessel workcenter occupation per volume per hour. While this improved the understanding of route processing costs, it did nothing to improve the understanding of materials and waste impact on the cost of goods or environmental metrics used in route selection.
Better Way
To improve the data model used in commercial route selection, Pfizer tested an alternative solution for knowledge management, based upon an upgrade in analytical capabilities and the flow of work and information. The analytical capabilities were developed using Aspen Technologies’ Batch Process Developer. Other improvements in information flow made the improved analytics more powerful than the previous workflow could have been. These included the use of electronic laboratory notebooks and electronic materials sourcing databases accessible to everyone on the co-design team.
The electronic lab notebook data capture and workflow meant that the scientists responsible for assembling process development cost and environmental information had a central repository for data. Within that environment, a continuously-updated structured, step recipe model was maintained for the purpose of easing technology transfer and enabling cost and green chemistry metrics analyses. This approach made it easier to collect detailed material consumption data and project stepwise operating cycle times on manufacturing scale. Centralized materials databases with historical price information further improved the level of detail in the materials data used in the co-design analysis.
Using this refined cost of goods and environmental analysis, Pfizer captures process development and scale-up history in a single platform rather than in multiple, customized spreadsheets. Each route is represented in graphical, synthetic flow for ease of editing. For access to project history, a list of established routes is maintained adjacent to the synthetic flow. After choosing a route of interest and displaying it as a synthetic flow document, users open the route and edit step inputs, within four categories describing the route: 1) properties; 2) materials; 3) processing; and 4) comments.
Properties input includes current and optimized step yields, which are based upon current experience in development and scale-up, plus expected improvements from long-term implementation. The materials section allows input of the full list of starting materials, reagents, and solvents—including the purity and mixture composition, if applicable—used in a particular step plus details about volume, mass, or mole ratios. The cost of each material is displayed as a line item from a separate materials database. Next, the user chooses a waste category for each material, in order to generate green manufacturing metrics with the standardized output.
Once all steps in one route are populated, the graphical synthetic route is automatically updated with primary information about each step, including the step intermediate name, molecular formula, molecular weight, current or optimized yield, and cost per unit mass of the intermediate and final product of the route. The cost basis can be set to include any combination of materials, processing, and waste disposal.
Pfizer finds this method of entry convenient, and transparent to the various users on a project team. Quick data entry into routes under consideration has made a significant difference in co-design discussions between research and manufacturing. Assumptions are often adjusted and displayed in route selection meetings for all project team members to view and evaluate.
Route Modeling Output and Analysis
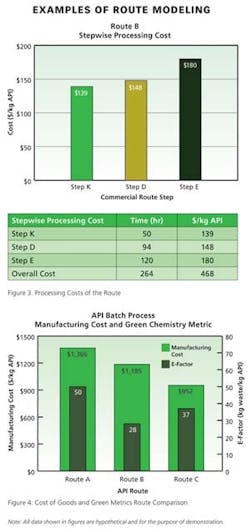
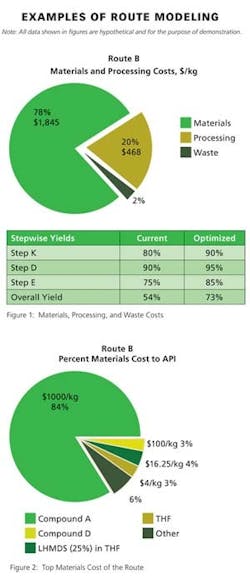
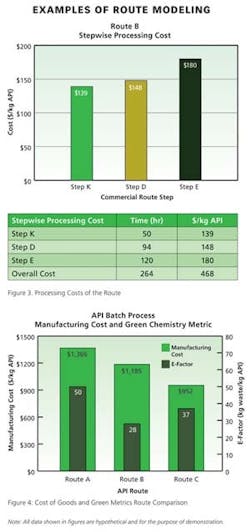
Beyond the primary data displayed directly in the process development software, detailed analysis of any given route can be produced as an Excel report based on a standard template. The co-design team finds this output particularly useful in examining details about materials, processing, or waste disposal within a particular step or the entire route. It is from this multi-page Excel report that Pfizer generates numerous graphics (Figures 1 – 4, for example) for use in co-design brainstorming sessions.
For example, the waste material summary relies upon information about the fate of each material in the stepwise material tables—waste sources are split by category and total waste is calculated per unit mass of active pharmaceutical ingredient, or product. The division in waste category information enables the project team to easily calculate three primary green manufacturing metrics: 1) the E-factor (waste mass/product mass), 2) the mass intensity (waste mass except water/product mass), and 3) the reaction mass efficiency (mass of product/mass of reagents). Other metrics of interest derived from this output template include the overall yield, number of isolations, mass of solvent except water/mass of product, mass of water/mass of product, mass of reagent/mass of product, and mass of starting materials and reagents/mass of product.
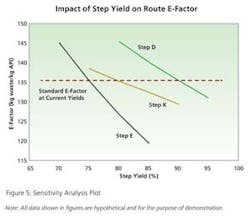
A comprehensive data summary is generated for each route in terms of cost of goods and green metrics. Members of the project team then have time to assess the data and draw preliminary conclusions before meeting to discuss and compare route options.A final component of route selection is a sensitivity analysis of various cost and environmental factors. One or more changes are made to key inputs in the interface and results are plotted for a visual indication of metrics sensitivity (Figure 5). Such sensitivity plots provide a means of prioritizing further process development or optimization of the chosen route in manufacturing. In regards to green manufacturing metrics, this could be improvements in step yields, reduced loading of reagents, or increases in processing concentration.
Benefits of a New Co-Design Approach
Co-design between R&D and manufacturing at Pfizer has been greatly enhanced through this centralized approach. It enables decision-making transparency within a project team, allowing its members to examine each step of each route under consideration thoroughly, for commercial nomination. The approach has improved the organization of data and results, which has fostered greater collaboration between divisions involved in route selection. Most importantly, Pfizer can better select synthetic routes that will have the greatest cost and environmental advantages in commercial production.
About the Authors
Eric Cordi has 15 years of experience as a research engineer in the pharmaceutical and crop protection industries. He is currently with the Engineering Technology group of Pfizer Chemical Research & Development, involved in multidisciplinary process development, design, modeling, cost analysis and technology transfer. Eric received degrees in Chemical Engineering from the University of Virginia (BS), Texas A&M University (MS), and University of Colorado (PhD). He also holds an MBA from the University of Colorado at Colorado Springs.
Russell Schofield has been with AspenTech for over 21 years, the past 15 as a technical support consultant with pharmaceutical companies, focusing on modeling and simulation projects. He previously held chemical engineering and plant management roles at Ciba Geigy and Cargill. Russell holds a BSc Honours degree in Chemical Engineering from Loughborough University.