In response to market forces and competitive imperatives, pharmaceutical manufacturers are consolidating manufacturing and packaging into fewer and fewer plants to reduce costs. At the same time, the number of unique packages required to meet market and regulatory requirements is increasing. These trends are rapidly rendering obsolete the traditional approach where pharmaceutical products are packaged and labeled at the time of production.
There is a more efficient approach to traditional labeling/packaging operations called bright stock manufacturing in which products are produced in large efficient batches and stored in unlabeled individual containers. These containers are then labeled just prior to shipment. The main challenge of implementing a bright stock manufacturing strategy is ensuring that the contents of every container are known with 100% certainty before applying the label and having the ability to demonstrate this fact via the pharmaceutical manufacturer’s quality assurance, quality control (QA/QC) regime.
NEED FOR JUST-IN-TIME LABELING
The pharmaceutical industry’s strategy of consolidating manufacturing operations into a smaller number of high-volume plants is challenged by the need for these plants to distribute its products effectively and reliably deliver shipments to meet the varying demand from local and global markets. To meet such market diversity and complexity each product requires an increasing number of unique labels to meet the regulatory and societal requirements of a given country or region.
The traditional approach of labeling and packaging the product at the time of production is becoming increasingly untenable because manufacturers are forced to estimate the demand for each of the different labels applied to its products. This approach usually requires that production be in relatively small batches, which is less efficient and more costly to produce than larger batches.
For example, when market demand doesn’t match the forecast, product rapidly loses its value as it moves closer to its expiration date. Even while labeled product is losing value sitting on the shelf, a given manufacturer may be forced to produce and label the same drug to meet unexpectedly high demand for the same product with different labels.
Pharmaceutical manufacturers have long wanted to move to the more efficient approach of producing bright stock in large efficient quantities and labeling and packaging the product just prior to shipment—when demand is known for certain. But making certain that the contents of each and every bottle match its label on application is a significant technical challenge.
How can the manufacturer be certain that a handful of bottles containing a different product haven’t been mixed together with the batch being labeled? Just a single bottle with the wrong contents could have enormous consequences including harm to a patient, damage to the manufacturer’s reputation, a multimillion dollar recall and so on. This same concern, it should be noted, also applies to the increasingly large number of pharmaceutical manufacturers that due to plant consolidation are now making products in one facility and shipping them to another for labeling.
VALUE OF MARKING EACH CONTAINER
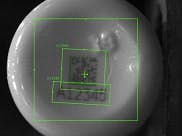
Processed image of 2D Matrix and human readable code
An obvious solution to this problem is to mark each container at the time of production to eliminate any doubt as to its contents. This code is then read just prior to applying the label and packaging in order to ensure that the bottle contains exactly what the label says it does. This raises the challenge of generating a mark on each bottle, often made of high density polyethylene (HDPE), recognizable with certainty during downstream processing. Traditional marking solutions such as CO2 and fiber lasers cannot generate much contrast on HDPE materials, rendering bar codes nearly unreadable. Ultraviolet (UV) lasers can generate a darker mark but it is still challenging to maintain the high read rates due to the many variables in the marking and reading process.
These unavoidable limitations in the marking process make machine vision critical to the success of just-in-time labeling. The traditional approach to machine vision involves the use of cameras controlled by personal computers (PCs). But PC-based vision systems take considerable time and effort to integrate because they require considerable vision expertise and in many cases knowledge of low-level programming languages. PC-based vision systems also require IT department oversight, service pack updates, and other items like virus protection software. PC-based systems can also be expensive in today’s regulatory environment because each upgrade to the PC and vision code may require revalidation to meet FDA requirements.
ADVANTAGES OF VISION SYSTEMS
FP Model 1371 Coding Machine
FP Developments Inc., a supplier of packaging machinery, recently provided a major pharmaceutical manufacturer with seven packaging lines. Each line deploys FP Model 1371 coding machines that mark a unique code on each bottle during the filling process and use a vision system to verify the code before the product is stored or transferred.
At the heart of this process are machine vision technologies engineered by Cognex, integrated with the coding machines and configured to read the relatively low contrast laser-marked 2D Data Matrix codes with near 100% read rates.
Controlled by internal microprocessors Cognex’s In-Sight vision systems operate independently of PCs. The security and recipes for the vision systems can be downloaded direct from the network or an industrial PC. Typically only one industrial computer per line is needed regardless of the number of vision systems to manage the security and recipes for the vision systems.
Vision systems are less expensive to implement because they can typically be developed without writing a line of code, a process replaced by a few prewritten functions called vision tools. The seven systems that FP Developments recently provided to a major pharmaceutical manufacturer use only two vision tools to read 2D Data Matrix codes marked with UV lasers on HDPE bottles. According to FP Developments Inc., Cognex vision systems are easily maintained by operators and easy to troubleshoot or perform routine maintenance. Operators can adjust the focus or lighting on the vision system either by plugging in a laptop or by operating the vision system in teach mode. Validation expenses are lower for vision systems because they are simpler and inherently more stable over time.
Cognex model 5600 integrated in FP Model 1371 coding machine
The vision system and laser are connected via Ethernet to a programmable logic controller (PLC). The PLC triggers the laser and vision system. The vision system compares the code on the bottle to the recipe and sends a pass or fail signal to the PLC. The system is currently operating at up to 250 bottles per minute with near 100% read rates and is capable of operating at substantially higher speeds.
PRODUCTION ADVANTAGES WITH ECONOMIC BENEFITS
FP Developments Inc. says its pharmaceutical customer is attaining the production targets and cost efficiencies set by its bright stock manufacturing strategy made possible by its new packaging lines. Now able to process larger batches, the company is experiencing cost savings and high service levels while reducing inventory and product obsolescence. Perhaps more importantly, the pharmaceutical manufacturer can prove that it has control of its labeling process and the means to virtually eliminate mis-packaging.
This approach also has the potential provide a solution to other production problems. For example another manufacturer had a minor problem with graphics in a small percentage of a batch. It ran the batch back through the packaging machine and used the vision system to scan each bottle and pull out the bad ones. In the event of a batch specific recall, the system could also be used to scan the recalled bottles to determine what percentage of the bad batch has been recovered and possibly to release the good bottles back into the supply chain.
CLARITY FOR COMPLIANCE
Regulators around the world have been working for years to develop a standard for a secure electronic record of the chain-of-custody of pharmaceuticals, often called ePedigree. The new packaging systems installed by FP Developments Inc. position its customer to achieve compliance with these upcoming regulations designed to prevent counterfeit drugs from making their way into the hands of consumers. The infrastructure needed to validate the origin of a unit package of pharmaceutical is not yet in place, however, pharmaceutical manufacturers that mark and read individual bottles have the ability to easily track the origin of each bottle. All in all, the new generation of serialization solutions based on vision systems can help pharmaceutical manufacturers reduce costs and improve the security of their supply chain.
For more information, contact Cognex, One Vision Drive, Natick, MA 01760-2059 USA. Tel (Toll Free): 1-877-COGNEX1 (1-877-264-6391), Fax: +1 508 650-3344, Email: [email protected],Web: http://www.cognex.com/