Continuous Powder Blenders for Pharmaceutical Applications
The only constant in today’s world is change, and this could not be truer for the current state of pharmaceutical manufacturing. “Right now the manufacturing experts from the 1950s would easily recognize processes today. In 25 years these same processes will be obsolete,” said Janet Woodcock, director of the Center for Drug Evaluation and Research (CDER) at the U.S. Food and Drug Administration in her keynote address at the AAPS annual conference in 2011. Continuous manufacturing will be a significant component in the future of pharmaceutical manufacturing.
The advantages of continuous manufacturing over batch manufacturing are well established. When properly implemented, continuous processes are almost completely steady, can be designed at scale, and can be used reliably to minimize segregation and agglomeration of ingredients. Moreover, a continuous process is also the perfect scenario for implementation of Process Analytical Technology (PAT) methods, which are required to ensure closed-loop control of the continuous process. The business case for continuous manufacturing is very robust. In product development, continuous manufacturing systems allow the user to perform complex DOE matrices in just a few days, and using a tiny fraction of the material required to perform a comparable study in batch mode [1] [2], thus enabling enormous savings in labor, analytical costs and capital [2].
If recent investments by large pharmaceutical companies in researching and developing continuous processes for manufacture of solid oral dosage forms are any indicators, then we are very likely see rapid change in this arena, taking much less than 25 years.
Interestingly, some of the unit operations that are involved in the process of making tablets and capsules today are intrinsically continuous in nature, namely, milling, spray drying, roller compaction, extrusion, capsule filling, tableting and packaging. This implies that an existing batch process can be transformed into a continuous process with relatively minimal change in most processing steps. A crucial unit operation that is usually practiced in “intrinsically batch” mode, and that captures the essence of the batch approach is powder mixing. The typical batch powder mixing process involves the addition of all ingredients to a large bin, double cone or V-blender, and the vessel is then tumbled for a predetermined number of revolutions to enact a time-dependent process, where the challenge is to determine a suitable end point. Similarly, wet granulation is performed in batch mode using either a high shear blender or a fluid bed system (both batch). Irrespective of the manufacturing route (direct compaction or granulation), powder mixing is involved in all manufacturing routes, often multiple times in the same process. In addition, pharmaceutical powders often display segregation and agglomeration tendencies as well as poor flow properties, and therefore the development of robust and reliable powder mixing processes is one of the main challenges in batch process development.
EXPECTED TO BE CHALLENGING
Given this situation, the development of continuous powder mixing processes ought to be expected to be challenging as well. Surprisingly, this is largely not the case, and a rapidly growing body of experience indicates that continuous powder mixing processes are much more reliable, easier to develop, and easier to monitor and control than their batch counterparts.
Continuous granular mixing is not really new to the process engineering community. Continuous mixers have been widely used in the food, mineral processing, detergents and the catalyst industry for decades. However, in a recent review on continuous powder blending by Pernenkil and Cooney, the following statement highlights the state of continuous mixing for pharmaceutical applications: “It is interesting to observe that pharmaceutical powders have not been reported in continuous blenders.” This is understandable because very few powder mixing processes used in the pharmaceutical manufacturing today are continuous and hence the dearth of published research in the area. However, in the past decade or so, both the pharmaceutical industry and academia have begun to investigate and publish on continuous powder blenders with pharmaceutical materials.
The objective of the article is to provide a brief overview of the current state of technology of continuous blenders for pharmaceutical applications. The article focuses on a popular class of blenders, herein called tubular blenders. Four tubular blenders are reviewed in detail, with a description of their physical characteristics, capabilities and general performance. Other common continuous blenders are described in brief to provide a broad view of the state of the technology. The article concludes with the authors’ view on the main open scientific questions in continuous powder blending, seeking to identify areas where future efforts could be useful.
Portillo et al.(2008 and 2009) characterized in detail two continuous blenders that were built by GEA. The first blender was fitted with 2.2 KW motor and rotation rates of the impeller of range from 16 rpm to 78 rpm. The mixer is 0.74 m in length and 0.15 m in diameter. An adjustable number of flat blades can be fitted in the mixing zone of the blender. The powder is discharged through an outlet weir, which can be fitted with screens of desirable holes. The size of the holes controls not only the size of the largest agglomerate exiting the blender, but also the powder holdup in the blender, which in turn dictates residence time and thus mixing performance. It was found that the residence time and number of blades passes experienced by the powder was strongly affected by the impeller rotation rate and the processing angle. The longest residence time resulted from upward processing angle and low impeller rotation rates, which also produced superior mixing performance.
The second blender was smaller, with a diameter of 0.05 m and 0.31 m in length. It can be fitted with a maximum of 14 blades. The blender is identical to the one discussed above, except for geometrical differences and the ability of the impeller to rotate faster compared to the first blender. Results obtained from a DOE-based study for the blender exhibited similar results compared to the first blender. The inclination of the mixer and the rotation rate were found to be the most significant parameters affecting mixing performance.
Another blender that belongs to the same family is the Gericke GCM 250 tubular blender (figure 1) which was characterized by Vanarase, et al. The blender is 0.3 m long and 0.1 m in diameter. The impeller can be fitted with 12 triangular-shaped blades equally spaced along the axis of rotation. The first and last blades are different from the rest of the blades. The angle of the blades with respect to the shaft can be changed to control the rate of “back mixing,” more properly quantified in terms of the axial dispersion coefficient of the flow. The mixer is equipped with a weir whose angle can be adjusted to provide control on the mass holdup. The angle of inclination of this blender cannot be easily adjusted, and thus the impeller rotation rate and angle of the weir are only process parameters that can be used to control the residence time. It was observed that the mean residence time decreased with increasing rotation rates, but the rate of axial dispersion also increased. Therefore, intermediate rotation rates exert the highest number of blade passes, thus maximizing strain and homogenization and resulting in optimum performance of the blender.
Another blender in the tubular blender family is the Glatt GCG -70. This blender can be retrofitted with spray nozzles and can be used as a shear granulator. The blender is 0.73 m long and has a diameter of 0.08m. The blender has 24 evenly spaced blades along the length of the tube. Equipped with three different types of blades, and an infinite number of blade angles to choose from, there are an unlimited number of configurations a user can setup in search for an optimum combination. This blender also comes equipped with an “angled” blade that contributes to back mixing. Finally, the angle of the discharge port can be adjusted to control the mass holdup in the blender.
In studies performed to characterize this blender, it was found that the blade shape had complex effects on the outcome of the blending process. Smaller blades tended toward high holdups and longer mean residence times. The angled blade had little effect on the holdup, but it appeared to be more effective at moving powder along the axis, thus affecting the residence time distribution. Various blade configurations examined had small, if any, effects on the quality of the radial mixing in the system. They did affect, however, the quality of the axial mixing.
TOTALLY TUBULAR
The blenders outlined in the table belong to the tubular family of blenders characterized by their mixing mechanism and mode of convective transport. The mixing mechanism in tubular blenders can be categorized into two separate modes: radial and axial. The material enters the mixer through the mixer inlet, and the powder crosses the pathway of several impeller blades attached to the central rotating shaft placed along the axial length of the horizontal cylinder (mixing zone). Convection transports the powder along the vessel axis (from the entrance to the discharge), erasing fluctuations in composition introduced by non-uniformities in the feed rate of ingredients.
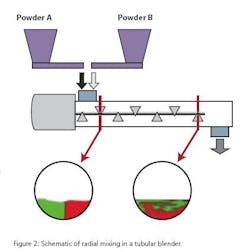
Convection is also the primary source of the second mode of mixing, cross-sectional (or radial) mixing, driven by the impeller blades. Consider two powders, A and B, being fed into a blender each on opposite sides of the blender’s axial center line at a constant speed. A snapshot of a radial cross section of the blender tube near the entrance at steady state would show two unblended powders.
If the blender is properly designed and operated, a snapshot of the radial cross section near the exit of the blender would show the two powders blended together. The key aspect of radial mixing in a continuous system is that it is a steady-state process, and can largely be considered time-invariant. As the blades rotate, a shear stress is applied, powders experience extensive strain and agglomerates are broken up. Particles collide with each other, or experience micro-avalanches, leading to stretching of unmixed areas and homogenization.
The effectiveness and performance of tubular mixers depend on how the aforementioned phenomena take place. Although this depends also on the material properties of the ingredients being mixed, the mixer performance can be maximized by choosing the correct design configuration (the impeller and blades). The number of blades, their configuration and angle are parameters that are found to be statistically significant in terms of their effect on mixing performance. The configuration of the outlet (weir angle/screen size) are additional parameters that control the material holdup in the blender, which dictates the amount of time the material spends inside the blender for a given flow rate and therefore the number of blade passes experienced by the powder as it travels through the mixer, and hence, the overall effectiveness of the process.
While at first inspection axial mixing may not seem as relevant as radial mixing, it is necessary to recall that the granular nature of powder products makes steady feeding very challenging. In fact, all powder feeding technologies will exhibit some level of variability in feed rate. If these variations were allowed to propagate down the manufacturing line, they would lead to variations in the end product composition. Therefore, the blender must be configured to incorporate enough axial mixing to dampen out the noise associated with powder feeding. Typically, high frequency fluctuations from the feeders are easily smoothed by the back-mixing action of the blender, while low frequency fluctuations are much harder to eliminate. A simple but useful rule is that the residence time in the blender must be at least five times larger than the characteristic time associated with the slowest fluctuation that the mixer needs to eliminate.
The blenders described have not been characterized by the authors group but are popular blenders in the market today.
The Readco Continuous Processor is a horizontal twin shaft, co-rotating, overlapping designed unit capable of mixing a variety of ingredients from dry powder and dry granulation to wet mass granulation in the same unit. The design of the mixer allows to changethe arrangement of paddles and screws inside the unit to provide control of the mixing and shear rates to suit the application. The unit has the ability to perform granulation, mixing, compounding, kneading, shearing, reacting and polymerization by the use of appropriate elements in the unit. The unit is virtually self-cleaning and can also be disassembled to perform more though cleaning if necessary. The manufacturer also provides stainless-steel versions of the paddle and screw arrangement if the configuration is being intended for long term use.
The Hosakawa Vrieco-NautaModulomix manufactured by Hosokawa Micron B.V., the Netherlands, is a continuous mixer/granulator based on the Cyclomix technology. By varying the rotational speed and design of the agitation impellers, different mixing and granulation conditions can be created. When a greater capacity is required, several Modulomix units can be connected in parallel. Alternatively, by connecting several units in series, multiple mixing steps can be combined; for example, mixing of a filler and an active ingredient in the first step, followed by the addition of a lubricant before the tablet press as a second step can be implemented.
Rahmanian and Ghadiri characterized the Modulomix to investigate the ability of granulator to perform seed granulation. They compared the granulator to Extrudomix(manufactured by Hosokawa Micron, UK) and Nica M6 Turbine (manufactured by GEA, UK) and showed that the Modulomix provided better performance in terms of its ability to produce seeded granules. This was attributed to the ability of the Modulomix to emulate the high shear environment needed for seed granulation.
It is clear from the above discussion that the design and operation of continuous blenders is relatively well understood (better, in fact, than for batch blenders). However, comprehensive process understanding is critical to minimizing risks during operation. Few works cited above have considered the material properties of the incoming material in evaluating performance of the blenders. Researchers have worked with common pharmaceutical excipients such as lactose and microcrystalline cellulose and active ingredients such as acetaminophen and caffeine. Although fine grades of these ingredients pose nontrivial mixing problems, formulations can be very complex, displaying multiple minor ingredients and more than one API, and it is therefore important that the performance of continuous mixers be evaluated for powders with a wide range of properties.
Micronized APIs are often used since they tend to enhance the dissolution rate of the active ingredient. However, the highly cohesive nature of these powders results in their tendency to agglomerate, ultimately leading to non-uniformity of the drug in the final product. While granulation is often an effective approach for dealing with such issues, the ability to apply a controlled amount of shear during continuous blending also enables the use of simpler and less expensive dry processes for such materials. However, to achieve such advantages, feeding and mixing of such cohesive materials needs to be examined in detail.
An intriguing scenario that has not been studied is the blending of materials with large particle size (300 microns or more) that exhibit a strong tendency to segregate, leading to content uniformity issues in the final product. While a properly designed continuous process greatly reduces the extent of segregation of a blend, significant studies must be conducted in order to develop robust approaches for mixing such blends effectively.
CLOSED-LOOP PROCESS CONTROL
Another area that is also crucial to the development of robust continuous blending technology is closed-loop process control. The relative ease of implementation of control methodologies in intrinsically steady continuous processes is one of their greatest advantages over batch systems. However, to enable closed-loop control, the relationship between material properties, blending parameters and blender performance must be determined. Implementation also requires fitting the equipment with appropriate PAT tools, which must be not only accurate, but also faster than the process, in order to enable the measurement to be used for control purposes. Selection of the appropriate control strategy is equally important, given the fact that multiple control schemes of unequal effectiveness can be potentially implemented.
Finally, science-based methods for process scale-up must be developed. While in many cases continuous processes can be developed in the same equipment that will later be used for manufacturing, there are a number of situations, particularly when developing processes for new products, where it is important to minimize the amount of materials being used to develop the process. In such situations, availability of small pilot equipment that can be easily scaled up can be critical to success.
The approach to overcome all of the challenges is straightforward. They can be addressed with a moderate amount of time and effort, guided by prior experience. As this process unfolds, continuous manufacturing will grow quickly and will become a major component of solid dose manufacturing.