The emerging technologies that characterize Industry 4.0 — from connectivity to advanced analytics, robotics and automation — have the potential to revolutionize every element of pharma manufacturing labs within the next five to 10 years. The first real-life use cases have delivered 30-40 percent increases in productivity within already mature and efficient lab environments, and a full range of improvements could lead to over 50 percent reductions in overall quality control costs.
Digitization and automation will also ensure better quality and compliance by reducing manual errors and variability, as well as allowing faster and effective resolution of problems. Use cases have demonstrated a more than 65 percent reduction in deviations and over 90 percent faster closure times. Prevention of major compliance issues can in itself be worth millions in cost savings. Furthermore, improved agility and shorter testing time can reduce QC lab lead times and eventually lead to real-time releases.
While most of the advanced technologies already exist today, few pharmaceutical companies have yet to see any significant benefits. On one side, quality leaders struggle to define a clear business case for the technological changes, and thus fail to bring to management attention to the significant impact potential associated with lab digitization or automation. On the other side, companies often neglect the development of a clear long-term lab evolution strategy and blueprint, which can lead to some costly investments with unclear benefits. For example, many companies have already taken steps to become paperless by first simplifying paper records to minimize the number of entries and then digitizing lab testing records. Now those moves are being superseded by new advances in equipment connectivity that enable direct transcription of thousands of data points without manual data transcription and without any reviews.
To capture opportunities offered by existing and emerging technological advances, companies should set clear goals, define robust business cases for any level of investment, and engage in rapid piloting of the new technologies followed by fast scale-up of pilots that deliver promising results. To succeed in the future, pharma companies need both the foresight to make long-term strategic investments, including those in R&D for developing and filing new test methods, and the agility to adapt those plans as technologies rapidly evolve.
THREE HORIZONS OF LAB EVOLUTION
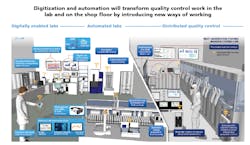
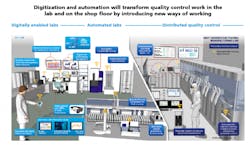
Multiple digital and automation technologies have created opportunities for change in pharmaceutical laboratories. Most pharma labs have not yet achieved digital transformation, but labs can aim for one of the three future horizons of technological evolution (see Exhibit 1).
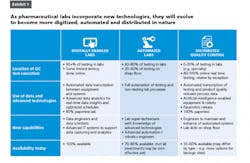
1.Digitally enabled labs achieve at least 80 percent paperless operations. These labs transition from manual data transcription and second person verification to automatic data transcription between equipment and general laboratory information management systems (GLIMS).
Digitally enabled labs use advanced real-time data analytics and ongoing process verification to track trends, prevent deviations or out-of-specifications, and optimize scheduling. They employ digital tools like smart-glasses to translate standard operating procedures (SOPs) into step-by-step visual guidance on how to execute a process. They create a digital twin of a lab to predict impacts before making physical changes. All of these are currently available technologies, with time-to-impact as short as three months for each case.
An average chemical QC lab can reduce costs by 25-45 percent by reaching the digitally enabled labs horizon. Potential savings at an average microbiology lab would be in the 15-35 percent range. Productivity improvements come from two main sources:
• Elimination of up to 80 percent of manual documentation work, and
• Automation and especially optimization of planning and scheduling to improve personnel, equipment and materials utilization.
With fewer manual errors and data-enabled analyses of root causes, labs can greatly reduce investigation workloads. The digitally enabled labs also reap compliance improvement benefits from reduced errors and variability, as well as seamless data retrieval and analysis. The increased productivity and scheduling agility can also reduce lab lead time by 10 to 20 percent.
One large global pharma company transitioned to a digitally enabled lab within its Italian digital lighthouse plant. Lab productivity at the site jumped by more than 30 percent after the company implemented advanced schedule optimization by harnessing a modular and scalable digital twin platform adapted to the lab specific scheduling constraints. The site also used advanced analytics to reduce deviations by 80 percent, eliminating reoccurring deviations altogether and accelerating deviation closure by 90 percent.
Pharma companies have many options when it comes to choosing and customizing technological solutions to create digitally enabled labs. In addition to custom digital twin and advanced analytics platforms, other solutions include real-time insights from IoT platforms, lab scheduling software, and digital assistants with visual operating procedures.
2. Automated labs use robots, cobots or more specific advanced automation technologies to perform all repeatable task, such as sample delivery and preparation. At the automated labs stage, some high-volume testing (e.g., microbial detection and water-for-sterility) is performed on-line instead of in physical labs. Automated labs can also use predictive maintenance technologies to plan for infrequent tasks like large equipment maintenance that can be performed by lab analysts with remote expert support.
While full implementation of digital enablement is not a prerequisite, automated labs can build upon digitization to deliver greater value and higher cost savings. Automated micro labs can enable additional cost reduction of 10-25 percent inside the lab, while also capturing a similar amount of savings outside the labs. The same improvements at chemical labs have the potential to produce 10-20 percent savings beyond that achieved by digitally enabled labs. The productivity improvements come from the automation of sample taking, sample delivery and sample preparation tasks, as well as from the reduction of equipment maintenance cost through remote monitoring and failure prevention. Automation also reduces sampling and related logistics tasks performed by operations outside the lab, which also produces cost savings.
Pharmaceutical companies can also achieve additional benefits beyond efficiency. Remote monitoring and predictive maintenance capabilities built into the equipment will decrease down-time and ultimately enable companies to reduce their use of expensive devices such as chromatography, NIR spectrometers, isolators, etc. By shifting to instantaneous microbial detection for environmental monitoring, companies may also reduce their overall lab lead-time by 40-75 percent.
3. Distributed Quality Control represents a true disruption to traditional ways of providing quality control. At these sites, nearly all routine product testing would take place on the production line, enabling real-time release testing (RTRT). Equipment and robots at distributed QC facilities have artificial intelligence capabilities. In the distributed QC scenario, labs continue to perform specialty and stability testing. This testing can take place off-site in a centralized location. Adoption of Process Analytical Technology (PAT) and RTRT has been relatively slow due to the regulatory filing and approval requirements. To be able to make a smooth shift to the on-line testing in the future, operations need to start collaborating with R&D now to develop an optimal quality control and filing strategy, especially for new products and manufacturing sites.
Distributed QC facilities primarily add value by reducing the footprint and costs of a traditional lab. Due to significant R&D investment requirements, as well as the need for equipment and operational changes, existing sites with stable or declining volumes are unlikely to make a compelling business case for distributed QC in the short and even medium term. At the same time, sites that have been rapidly growing or under construction may be able to capture value from reducing capex investment for building or expanding traditional QC labs if they can move significant share of the routine testing on-line. Distributed QC and real-time release would also enable true continuous manufacturing processes.For example, Biogen plans to use this distributed QC method of real-time release and review by exception in its new manufacturing facility near Solothurn, Switzerland.¹ When production starts in 2019, the Solothurn facility will achieve raw material control through screening and genealogy, with minimal testing using Rapid ID and electronic data exchange. Bioreactor processes controlled through inline instruments will eliminate the need for process control sampling. The facility will have adaptive process control levers, lab execution by recipe, and automated data transcription from all equipment, all based on an understanding of raw materials, processes and product characteristics. The integrated control system allows employees to see data and react in real time.
TYPICAL IMPLEMENTATION PITFALLS
As pharma labs evolve, they face significant costs associated with implementing IT and automation solutions. Even expensive solutions can deliver strong positive ROI, but many companies, unfortunately, struggle to capture value from these digital upgrades. These companies typically encounter one or more of the following pitfalls:
Not having a clear vision of what evolution horizon is the right target for a specific lab: While most labs can make a solid business case for the digitally enabled horizon, not all labs have sufficient volumes and operational setup to justify automation and distributed QC. For example, it could be hard to justify an investment in automating a smaller lab where the potential cost savings might be less than $200,000 a year, whereas the same investment could quickly generate positive ROI for a large sterile facility with significant environmental monitoring volumes.
Not having a compelling business case for the transformation: Many companies start the implementation of costly IT systems without a clear understanding of the full benefits such solutions can generate. This often results in delays in implementation and the roll out of partial solutions. For instance, labs might move to paperless systems on individual modules, but still need significant manual efforts to move data from one system to another. This can lead to situations where analysts must record test results into a paper log before manually entering the data into a laboratory information management system (LIMS). This manual entry step prevents them from capturing the full savings they should get by from automating documentation.
Targeting to have a fully tested end-to-end future state prototype rather than testing and rapidly scaling up high-value solutions to capture quick wins: For example, scheduled automation and optimization can be quickly implemented and start generating significant value even if a lab is not yet mostly paperless and fully digitized.
Lacking proper planning or management for rollout of new systems and technologies: In extreme cases, it can take pharma companies several years and more than $100 million to implement a LIMS. Given such a lengthy timeframe and the fast pace of technological change, some of the LIMS capabilities are liable to become obsolete before they get rolled out across the entire network. Pharma companies need skilled resources to accelerate the rollout and should avoid the temptation to engage in excessive customization at each site. A poor rollout can cost up to 10 times more and take much longer than a properly planned investment executed with good long-term planning.
Not having a full understanding of the capabilities of the systems they acquire: Pharma companies may purchase a system such as LIMS to comply with data-integrity regulations without truly understanding or considering the system’s potential to generate improvements in productivity.
Pursuing automation rather than optimization: Scheduling automation can deliver two-three percent of the QC cost savings, but automation plus dynamic scheduling optimization can yield three to four times more value.
Self-imposed constraints from perceived need to validate all systems and technologies: Many of the high-impact changes such as optimized scheduling and data-enabled deviations analysis do not require validation and refiling.
Missing the skillset to extract full value from their data: Most typical pharma labs do not have the advanced analytical capabilities needed to get the maximum value from data sources. As a result, the labs collect data, but it does not get used properly to generate insights that could prevent problems or reduce testing volumes.
Spending too little time and effort on developing a robust change management program: Digital transformation requires radical changes in mindset and has major implications for the organization and individual employees who must develop new skills and competencies. To succeed, companies must make upfront investments in changing the culture, winning buy-in across the business, and forging strong links between business and IT functions.
HOW TO GET STARTED
The good news is that most of the technologies needed to attain any of the three horizons of Industry 4.0 QC labs already exist. Many of the technologies mentioned are currently being deployed in pharma environments, with some successful pilot projects already completed and others in the approval stage.
To successfully implement Industry 4.0 technologies, pharma companies need to set the right aspirations and move quickly. Here are key steps to get started today:
1. Test several use cases and technologies quickly to find the best ones for each lab type.
2. Create lighthouse QC Labs to showcase the potential benefits of amalgamating these innovative technologies.
3. Find out which innovative tools can have the greatest immediate impact, then roll them out quickly across multiple sites. Don’t get bogged down trying to set up a fully functioning lab with every possible desirable technology. Many use cases such as scheduling optimization can be implemented before other elements (e.g., paperless labs) are in place.
4. Establish a clear target state and business case for each lab early on. Track the value capture along the way and reinvest the savings toward the next 4.0technological upgrades. It is important to make an assessment separately for chemical and micro labs, because the baseline cost and the impact of improvements may differ significantly.
5. Aim for the highest value horizon justified by the business case when planning and building new labs to preempt the need for digital transformation upgrades right after the lab opens its doors.
6. Start building the needed talent base and skills early on. Clearly understand future capability needs and invest in training high-potential employees and hiring employees with the new required skill sets (e.g., advanced data analytics) during early stages to enable faster scale up.
Modern technologies can make QC faster, more agile, reliable, compliant and efficient. By setting appropriate goals, choosing the right technologies, and scaling up quickly, pharma companies can become QC leaders and reap the rewards in the form of speed, compliance, cost savings and productivity improvements.
REFERENCES
1. Biogen presentation, EMEA Users Conference, OSIsoft, October 2017, London, United Kingdom.