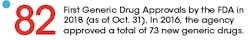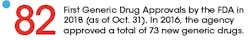In a year marked by rapid technological shifts and mounting political pressures to solve pharma-related policy issues, U.S. regulators have had their work cut out for them. But at the start of the year, the U.S. Food and Drug Administration’s commissioner, Scott Gottlieb, laid out a policy roadmap for the year that promised to tackle some of the country’s thorniest health troubles.
By and large, the agency has delivered on its promises, crafting a range of new guidance documents and regulations that address critical pharma policy areas such as opioid addiction and prescription drug costs.
But the important question is: What does all this mean for pharma manufacturers?
It’s no secret that staying on top of regulations is a daunting but necessary endeavor for the pharma industry. To help navigate the shifting landscape of pharma regulations, we’ve put together an easy-to-follow guide of the most noteworthy changes in 2018.
The new policies target everything from drug discovery and approvals to facility inspections. Other changes, such as those related to opioids, represent a cultural watershed for the industry, that could be felt for decades.
Here are the biggest hits from 2018 and what you need to know to maintain your company’s regulatory intelligence.
Prescription drug prices continue to be one of the most vexing issues facing the industry. President Donald Trump has been notably outspoken on the issue, using hostile language toward “Big Pharma” and promising to drive costs down.
Meanwhile, the FDA strategy for addressing the issue has been to promote generics, with the goal of getting more of them on the market. In addition to streamlining the process of getting generics approved, the FDA has been working on ways to prevent manufacturers of brand-name drugs from “gaming” the system and blocking generics from entering the market.
GDUFA II
Back in 2012, the Generic Drug User Free Amendments (GDUFA) were passed as part of the Food and Drug Administration Reauthorization Act. For the first five years, the FDA put the pieces in place to achieve the primary goal of GDUFA: bringing greater predictability and a faster timeline for getting generics reviewed and approved.
Late last year, the plan was reauthorized (and is now known as GDUFA II), and the results of the previous five years are now being felt in the industry. From the perspective of Srinivasa Rao, vice president and head of Regulatory Affairs-North America Generics for Dr. Reddy’s Laboratories, a multinational pharma company that specializes in generics, the impact in 2018 was enormous.
“I’ve been doing this for 20 years, and I’ve seen a huge change with GDUFA II,” Rao says. “Back in the 90s, it might take an application for a new generic drug 20 to 25 months to get approved. Now, the timeline for a high-quality application could be as fast as eight to 10 months.”
To expedite applications, the agency increased its review staff, which meant it had to ratchet up the cost of application fees. Prior to 2012, there was no fee for applying for a generic drug approval. In 2018, however, the cost was as high as $172,000 for an Abbreviated New Drug Application (ANDA) and in 2019, will increase to $179,000. But Rao says that for Dr. Reddy’s, which files an average of 25 ANDAs per year, the extra fees are worth the speedier approval times.
“The FDA has given us more clarity through guidances and controlled correspondences,” he says. “In addition, there is another new approach called a Pre-Submission of Facility Information Related to Prioritized Generic Drug Applications, where you have the opportunity to get an expedited review and ANDA approved in eight months if you send the required data ahead of time.”
Created in 2017, the Pre-Submission option allows companies to submit accurate facility information in advance of their ANDA to help expedite review.
Overall, Rao says the changes have been a major win for consumers, and despite the increasing competition on the generics scene, GDUFA could incentivize more companies to enter the generics market.Global Harmonization
At total of 16 countries have now signed on as members to the International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use (ICH), along with 28 observer countries. The goal of ICH is to help create global standards for medicines, which could become a key driver in bringing more generics to the global market.
This past November, Gottlieb submitted a proposal to the ICH recommending the development of internationally harmonized guidelines on scientific and technical standards for generic drugs.
“The lack of harmonization across such basic components of generic drug development reduces the opportunities for generic drug developers to use their data and information across multiple applications in different jurisdictions. The result is increased cost and complexity to generic drug development,” Gottlieb said in a statement. “As a consequence, generic drug developers with limited resources sometimes must choose only a limited number of countries where they’ll apply for marketing approval.”
According to the FDA, Gottlieb’s draft Reflection Paper on the issue was endorsed by the ICH Assembly, and Gottlieb said the FDA will continue pushing for policies to create global, harmonized standards for generic drugs.
The Drug Competition Action Plan
Since its adoption in 2017, the FDA has used the Drug Competition Action Plan to push for policies that support the generic drug market. In 2018, the agency requested over $35 million from the Congressional budget to fund two major initiatives within the FDA. One will include the switch from its text-based generic drug review platform to an updated data-based assessment tool. This change will help the agency process generic drug applications in a timelier and more efficient manner.
The other initiative is aimed at helping the agency take more of an active role in keeping generic drug labeling up to date. Typically, innovator drugmakers update labeling information for the brand-name medication, which then applies to the generic. But if the innovator drugmaker takes the product off the market, the process of keeping the generic label up to date can slow down, which could result in the drug being prescribed less often. Keeping the labels updated will ultimately remove a barrier to having more generics on the market.
The CREATES Act
Although it hasn’t been passed yet, the Creating and Restoring Equal Access to Equivalent Samples Act has mostly received broad bipartisan support and is making its way through legislative channels.
According to supporters, the impetus behind the bill is to stop brand-name companies from abusing the FDA’s Risk Evaluation and Mitigation Strategy (REMS) program to keep samples out of the hands of generic drug developers. If adopted, the CREATES Act will allow generic drugmakers to bring civil lawsuits against brand-name manufacturers who fail to provide “sufficient quantities” of the drug samples needed for premarket testing.
At the time of writing, the bill was waiting for a vote in the Senate.
A New Tune for Drug Development
The need for speed hasn’t only been felt in the drug approvals stage — the FDA is also taking targeted steps to make early drug development more efficient.
Promoting Targeted Therapy Innovation
In October, the FDA released two new guidances aimed at modernizing drug development for targeted therapies.
The first draft guidance, “Hematologic Malignancies: Regulatory Considerations for Use of Minimal Residual Disease (MRD) in Development of Drug and Biological Products for Treatment,” is aimed at helping drug developers use MRD as a biomarker in clinical trials for specific blood cancer treatments. According to the agency, MRD assessments may be useful in clinical trials and have the potential to expedite product development.
The CDER Shakeup
The FDA also unveiled plans for a major reorganization that affects one-third of the Center for Drug Evaluation and Research (CDER). In an effort to make the process of working with FDA experts on early drug development more efficient, CDER director, Janet Woodcock, announced that the changes in the agency’s various divisions will impact how drugmakers are guided through clinical trials.
Project management for the Office of New Drugs will now be centralized, and various other divisions will be reduced and reorganized into clinical divisions. Ultimately, the goal is to have drug companies work with FDA experts more aligned with the type of therapy being developed.
According to Meredith Brown-Tuttle, a principal consultant with Regulatorium, the reorganization should eventually make the clinical trials process more efficient.
“It will be better, because I’ll have experts at the table who can talk to me about GI or liver disease, for example, who focus on nothing else,” she says.
Forever in Genes
The science behind gene therapies is evolving rapidly, but the FDA has been on the industry’s heels, and working to keep regulatory standards up to date. Recognizing the promise gene therapies hold for a swath of illnesses, including previously untreatable diseases, the agency released six new guidance documents in the first half of 2018 aimed at providing more clarity at gene therapy development and manufacturing.
Some of the new guidances deal with clinical trials (see above) and creating a clarified framework for using a genetic-based approach to early drug development. In fact, the agency has been notably lenient about allowing gene therapies into phase 1 trials. But when it comes to scaling up manufacturing, the agency has begun to develop clearer regulatory controls.
That said, the agency has also indicated that even though patient safety is, of course, paramount, the FDA also accepts some level of uncertainty when it comes to the burgeoning field of manufacturing gene therapies.
“In contrast to traditional drug review, some of the more challenging questions when it comes to gene therapy relate to product manufacturing and quality, or questions about the durability of response, which often can’t be fully answered in any reasonably sized pre-market trial. For some of these products, we may need to accept some level of uncertainty around these questions at the time of approval,” the FDA said in a statement in July.
In an emailed statement, the FDA said that its series of guidances from 2018 should “lay the groundwork for continued advancement by establishing a sustainable, evidence-based regulatory framework for future developments in this innovative field.”
Chemistry, Manufacturing, and Control Information for Human Gene Therapy Investigational New Drug Applications
This draft guidance is aimed at giving gene therapy developers recommendations on how to assure product safety, identity, quality, purity and potency with new gene therapies.
The 54-page guidance is organized with a similar structure as the Common Technical Document, and applies to gene therapies that are used in combination with another drug or device.
Long Term Follow-up After Administration of Human Gene Therapy Products
Because of the uncertainty involved with these novel therapies, the FDA has created guidelines for companies to follow when conducting post-approval trials. These standards detail which product characteristics, patient-related factors and clinical data should be studied to create the kind of robust, post-market follow-up needed to better understand the impact of new gene therapies, and help ensure long-term patient safety.
ReMixing Inspections
The days of FDA inspectors climbing into full-body Tyvek suits and carefully monitoring manufacturing facilities are far from over. But this year, the efforts to modernize and streamline the process reached a critical new turning point.
New Inspection Protocol Project
Since the New Inspections Protocol Project (NIPP) was passed in 2015, the FDA has been working to bring facility inspections into the digital age. According to the agency, NIPP aims to shift to data-based tools to “facilitate the collection of information during the inspection in a standardized format.”
“The electronic inspection protocols enable semi-automated generation of Establishment Inspection Reports for pre-approval and surveillance inspections…and are expected to save investigators time in compiling data, which will also help with efficient analysis of the inspection,” the FDA said in an emailed statement.
In 2018, the agency announced that NIPP was moving beyond the pilot testing phase and is now being implemented in certain facilities. The agency chose aseptic facilities as its first targets because of the higher safety risk involved in sterile manufacturing.
The FDA is now working on expanding the protocols to cover unit operations with other dosage forms and plans to have NIPP ready for full implementation by the end of 2020.
Much like the pharma industry as a whole, the FDA has been a target for criticism about its lack of action towards preventing the rise of opioid addiction. But in recent years, the agency has kicked its efforts to address the ongoing crisis into high gear.
In 2018, the FDA released a string of new regulatory standards in guidance documents that address various parts of opioid manufacturing and consumption with the goal of reducing the number of new addiction patients, treating current opioid addicts and continuing to support adequate relief for pain patients.
Leveraging REMS
In September, the FDA approved new rules that extend the Opioid Analgesic REMS guidelines for long-acting opioids to immediate release (IR) narcotics as well. The new rules will require manufacturers of IR opioids to provide training to healthcare professionals about safe prescribing practices for IR opioids and prompt them to consider alternatives.
Transforming Pain Med Development
In August, the FDA announced that it plans to withdrawal its analgesic guidance document, which was released in 2014. Calling the existing guidance too broad, the agency said it will be releasing a series of new guidance documents through 2019 that will push drugmakers in the direction of developing new treatments with a limited indication for a specific type of pain.
The FDA also launched an innovation challenge to spur development of medical devices and other alternative treatments to opioids.
A Final Note
This, of course, is just a glance at some of the high points from the year. Although the FDA had many regulatory accomplishments such as tackling opioid addiction and promoting generics competion and gene therapies, there is still much to be done. As 2018 comes to a close, regulators will have their hands full keeping pace with pharma innovations and promoting healthier, more affordable medicines in the year ahead.
[javascriptSnippet]