Sterilization Methods' Impact on Packaging Component Machinability
Pharmaceutical manufacturers are faced with numerous challenges during production, filling, packing and transport of pharmaceutical products to patients and care givers. Common concerns for all pharmaceutical manufactures include meeting targeted timelines to guard against drug shortages and ensure speed to market. Manufacturing delays can result from the stopping of filling lines, product/component line clearances and pending investigations, which can lead to reduced output of finished product. Maximizing filling line operating speeds and minimizing line down time are essential to handling these issues and facilitating manufacturing efficiencies for the benefit of all stakeholders.
Machine Setup
The capability of parenteral packaging components (containers, elastomeric components and caps) to efficiently flow and effectively be processed on drug manufacture filling lines is commonly known as the machinability. The machinability of parenteral packaging components is recognized to have a significant influence on productivity of the drug product manufacturing process. Following is a close look at elastomeric components intended for aseptic processing with respect to machinability on a lab scale filling line.Parenteral packaging components used in aseptic drug manufacturing are required to be clean and sterile when intended for drug products for human use.21 CFR 211.94(c) states that “Drug product containers and closures shall be clean and, where indicated by the nature of the drug, sterilized and processed to remove pyrogenic properties to assure that they are suitable for their intended use.” There are various processes that can be used for washing and sterilizing elastomers for aseptic fill, and depending on conditions of processing, the physical and functional characteristics of the component will be altered to some degree.
The two most common means of sterilizing elastomeric parenteral packaging components are steam autoclave and gamma irradiation. When appropriately validated, both processes are very effective at eliminating microbial contamination of components used for container closure of sterile liquid, lyophilized or powder drug products. Despite the ability of both methods to reach the sterility requirements, the effect on physical characteristics of elastomers (as it relates to machinability of the components) can be quite different. In an effort to understand the impact of the two sterilization techniques on machinability, a study was performed by West Pharmaceutical Services (West)and Bausch + Ströbel Maschinenfabrik Ilshofen GmbH+Co. KG (Bausch and Ströbel).The objective of the study was to assess filling line speeds relative to performance of steam sterilized plungers versus plungers sterilized at two different levels of gamma irradiation.
MACHINABILITY STUDY DESIGN
The machinability study was performed using plungers designed for a 1 mL long syringe. The plunger design chosen for this study was laminated with a fluoropolymer film (FluroTec barrier film) and coated with a cured silicone (B2-coating).The plungers were subjected to the same washing but different sterilization processes. West collaborated with Bausch and Ströbel to conduct a study using six plunger sample sets composed of two different elastomeric formulations (bromobutyl and chlorobutyl). Sterilization variables included: i) steam autoclave processing for one hour, ii) gamma irradiation at 10-20 kGy and iii) gamma irradiationat40-50 kGy. Both samples sets of gamma irradiated bromobutyl plungers were from the same manufactured, washed and B2-coated lot; however, the difference was the degree of gamma irradiation. Also, both samples sets of gamma irradiated chlorobutyl plungers were from the same manufactured, washed and B2-coated lot; however, the difference was the degree of gamma irradiation.
MACHINABILITY ASSESSMENT
Each sample set was evaluated on a lab scale filling line, which was equipped with a dosing trough, feeder bowl, sorting track and feed track. The filling line setup consisted of a 10-lane feed track with a maximum line speed of 600 plungers per minute. Four different line speeds were investigated to determine if the machining performance requirements were met. The performance was judged based on two criteria: a smooth consistent flow and lack of line blockages. The feeder bowl and tracks were designed by Bausch and Ströbel for this study and is shown in Figure 1. A high-speed camera was used to capture images of the plunger movement within the feeder bowl and tracks at each of the various speeds tested. The images were examined for areas of sluggish or undesirable performance during the test runs.
Machinability Study Results Overview
The same study criteria and parameters were used for each sample set test run. Those parameters were:
• Tested line speeds (as defined by plunger gripper settings):200, 350, 500 and 600 plungers per minute
• Quantity of plungers tested per sample set:25,000-30,000 plungers
• Run time per tested line speed: Approximately five minutes before incrementally increasing to the next higher speed
• Power setting for vibration solenoids of the dosing trough, feeder bowl, sorting track and feed track: Only one power setting (operation mode-full power) was used during the study.
The power setting was not reduced during the initial filling of the sorting and feed tracks, as typically is performed during drug product fill/finish manufacturing. By initiating the power setting at operation mode-full power, performance differences between each sample set were recognized/identified more clearly.
• Cleaning and set up activities following each sample set test run: Removed all remaining plungers from the filling line and dismantled all change parts for preparation of cleaning. Removed all residual silicone and macroscopic contaminants from the complete system (dosing trough, feeder bowl, sorting track and feed track). Once cleaned, the system was returned to the same previous operational setup.
MACHINABILITY PERFORMANCE
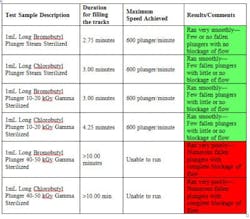
The study results showed that the steam sterilized plungers were consistently quickest to fill the feed tracks, and could achieve successfully maximum line speeds of 600 plungers per minute. The plungers gamma irradiated to 10-20 kGy were slightly slower to fill the feed tracks; however, they also could successfully achieve maximum line speeds of 600 plungers per minute. The plungers that were gamma irradiated to 40-50 kGy showed poor performance and could not meet the machining requirements at any of the filling line speeds, especially at 600 plungers per minute. Table 1 shows the overall results from the machinability study, as noted by Bausch and Ströbel.
Additionally, the plungers gamma irradiated to 40-50 kGy were observed by Bausch and Ströbel to exhibit a “stickiness” between the plungers sliding past one another as well as moving along the contact surfaces of the filling line. The hypothesis is that elevated levels of gamma irradiation, such as 40-50 kGy, can alter the elastomer physical properties, thus leading to tacky/sticky characteristics. In such a case, the most common means to overcome this phenomenon would be to increase the level of lubricating agent (silicone oil) applied to the elastomer. Since the level of silicone oil on the plunger takes into consideration several attributes of the final package, it was beyond the scope of this machinability study.
SUMMARY
Due to the overall importance of maximizing operating speeds and minimizing operating down time, the pairing of parenteral packaging components, drug product, processing and fill/finish equipment must be assessed thoroughly. As indicated by the plunger machinability results, the elastomeric parental packaging components used in aseptic drug manufacturing can be impacted by the sterilization method and conditions. It is the responsibility of the drug product sponsor to evaluate and understand suitability of packaging components during development, scale up and commercialization. Specific considerations should include understanding the impact of sterilization on component machinability.