Solvent Recovery at Pfizer: A Continuous Solution for Small Waste Streams
Solvents are often the primary waste company in API and drug processes, and solvent recovery is routine when it is environmentally and economically feasible. Pfizer has been recovering solvents for decades, and is continuously seeking ways to enhance that feasibility. From 2006 to 2009 at its Kalamazoo site, solvent recovery efforts have led to $65 million in savings, reports Frank Urbanski, Director of Engineering Technical Services, Pfizer Global Engineering.
Recently, Pfizer Global Engineering has worked with students and faculty at New Jersey’s Rowan University to research and develop green approaches to drug manufacturing, including solvent recovery. Led by Urbanski, scientists and engineers from Kalamazoo and Peapack, New Jersey, have explored solvent recovery as an alternative to incineration for small-volume waste streams.
Capital investment for such recovery typically includes piping, tank farms and recovery equipment, Urbanski notes—easy to justify for large volumes and expensive solvents, but not so easy with smaller streams that can’t necessarily be pooled with other solvents.
The researchers performed a case study on several API production waste streams in Kalamazoo, starting with the recovery of acetonitrile from the synthesis of selamectin (the active in the veterinary drug Revolution). Rowan designed a small-scale distillation, solvent-recovery system, and also evaluated it for recovery of isopropanol (used to manufacture nelfinavir, the active in Viracept) and toluene (used to make hydrocortisone).
We spoke with Urbanski to get more feedback on this project and other solvent recovery efforts at Pfizer:
PhM: What’s particularly challenging about solvent recovery from small-volume waste streams? It’s not just a matter of scaling down large-volume practices?
F.U.: The bottom line is it depends on whether your existing plant was designed to handle smaller quantities of solvents in an efficient manner. Certainly, if new equipment were to be purchased, the design can be scaled to the desired flow rates. If constrained to existing equipment, there would normally be some degree of turndown capability, allowing operation at a lower steady-state feed rate. Alternatively, a column could be operated for a shorter duration at steady state. Many times some cleaning will be required between different recovery processes, and more frequent changeovers for small recovery runs adds some degree of inefficiency. So, one might be able to operate an existing column if capacity is available, though less efficiently.
While Pfizer API plants are typically quite flexible, they are not designed to handle every scenario. Excess variability adds to plant cost. So, designs are based on assumptions regarding number of products, batch sizes, number of batches, etc. Similar assumptions would have been made to justify the incremental investment in solvent recovery assets, such as tank farm capacity (number and sizes), transfer lines, and recovery equipment (distillation columns, Pervap systems, etc.). Recovery of 100% of waste solvent streams would not be a typical design assumption and thus a facility would have constraints in the variety and volumes of solvents (virgin, recovered, in-process, etc.) that can be transferred, collected, stored and recovered at any given time. Pooling of solvents can therefore help by reducing the number of isolated streams that must be managed at an operation if it is possible for the particular solvent stream, and if the plant design, lab resources, process validation and quality systems are appropriate.
Solvent recovery operation is secondary to production in that it must react in response to changes in the production schedule, and continually seek to optimize asset utilization while trying to maximize benefits to plant finances. Small spent solvent streams tend to have lower value, and so receive lower priority in the scheduling of equipment, if there is capacity at all. When actual product mix strays from the design basis, and trends toward more variability in products, shorter campaigns, smaller quantities, or increased numbers of solvents, then fitting the solvent recovery demands to existing equipment becomes more challenging.
PhM: Do you think that small-volume waste streams are often overlooked by manufacturers as opportunities for solvent recovery, perhaps viewed as not cost effective or too much trouble? Has this project changed your/Pfizer’s thinking in terms of what is possible, and economically viable?
F.U.: In today’s environment, it is doubtful anyone simply overlooks a hazardous waste that is costing them significantly. Pfizer colleagues are engaged in serious continuous improvement, and reduction in waste volume is a target area. But, there are constraints as described above, and not all streams are being recovered—some because it is technically not possible, but others because capacity is not available or capacity does not fit the situation.
The idea for this study came up as I was thinking about how solvent recovery might operate if we had a continuous low-throughput API manufacturing process. I envisioned solvent recovery would be small scale and fully integrated with the manufacturing process, continuously being recycled, with small inventory in the system. There would not be a large collection tank for the waste stream somewhere out in a tank farm. Rather, the recovery column would be located very close to the manufacturing process, and the waste solvent stream would feed continuously, operated by the same technicians as part of the manufacturing team. The recovered solvent would be continuously monitored by PAT devices and fed back to the process, with little or no accumulation of recovered solvent in a large tank. I thought the same concept could be applied to small-volume batch mode API operations for streams that were not being recovered currently.
In contrast to that hypothetical operation, our solvent recovery facilities are typically separated from the API manufacturing building, and operated by different staff and on their own schedule, but in support of manufacturing. In a large plant, there could be dozens of collection and storage tanks, several recovery columns and sometimes a Pervap system. Waste solvents are recovered in batches, processed as a batch, collected as a batch, and then released as a batch. Only then, are they re-introduced to the manufacturing process.
So does it change my thinking? I think the study demonstrates that there may be situations in existing plants that could justify an atypical approach to solvent recovery for small volume streams when the mix of streams does not have a good fit in existing equipment. This is only one potential approach, which may be viable. Some of the considerations that would tend to make the approach more favorable are:
- Existing large-scale equipment is at capacity
- Small streams cannot be pooled; they need to be kept product-dedicated
- Relatively expensive solvents are being used once and discarded
- Multiple small-volume products are scheduled in series, the wastes from which are candidates for recovery
- Any new recovery equipment can be maintained near full loading
- A recovery column can be integrated with the API building operation to minimize new tankage and capital, solvent inventory, and manpower
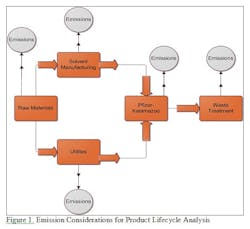
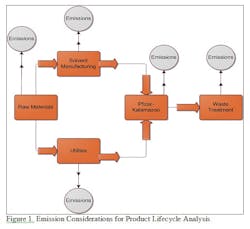
For example, if a Life Cycle Assessment approach were used to compare the impact of two organic synthesis routes for a molecule during process development, it could help scientists develop a “greener” manufacturing process considering much more information than simply the molecules in the synthesis. Pfizer is interested in further exploring the use of the LCA approach, and did sponsor another collaboration with Rowan in the 2010-11 academic year, using the LCA approach to situations beyond solvent recovery applications.
PhM: Can you elaborate upon how the initial acetonitrile recovery project is supporting work in other areas? Are the exact recovery system and procedures being used in other projects (e.g., isopropanol, toluene), or are they just serving as a model by which other systems may be built and operated?
F.U.: To clarify, the study completed by the Rowan team was a case study to look at the environmental impact and financial feasibility of investing in new, smaller scale equipment to recover small volume waste streams at the Kalamazoo site that were not being recovered. The economic analysis was not quite at a supportable level based on the identified potential recoveries, and thus the capital investment considered in this study has not been made. If additional recovery streams could be identified to utilize the 35% capacity that would be available, it would have more likelihood of support.
The good news is that in parallel with this work, the Kalamazoo plant was seeking expansion of its large-scale capability to recover chlorinated solvents via a new continuous recovery system. That project was approved, and will free capacity in an existing batch distillation area. The Rowan student team had simulated a recovery process options for the streams, proposing recovery conditions that should yield acceptable purity. Therefore, the plant is proceeding to utilize the freed-up capacity to implement “non-pooled” recovery of the acetonitrile from Selemectin, and isopropanol from Nelfinivar.
Expanding beyond the Kalamazoo site, the concepts and results of this study are being shared with Pfizer’s other API sites to determine if other opportunities exist.
PhM: Cross-contamination is always a concern in multi-product facilities which tend towards smaller waste streams—have you developed more sophisticated methods for the pooling of solvents from different streams? Could you provide an example or two?
F.U.: Pfizer has Quality Standards and Good Practices in place that define requirements for the recovery and reuse of solvents, including the practice of pooling. (Pooling is the combination of a solvent from various sources for use in various applications—i.e., not dedicating the use of a recovered solvent to the process from which they came). The risk of cross-contamination is addressed. Validation is required for solvent recovery processes if recovered solvents will be used in processes from which they did not originate. Solvents from processes that require dedicated facilities (e.g., penicillins) are not recovered for reuse in other product families. While pooling is a common practice at the Kalamazoo site, differences remain across our other API locations, primarily due to legacy company practices. We are moving towards pooling as an efficient practice where appropriate.
PhM: This case study, for three drugs, reduced carbon emissions roughly 677,000 kg/yr. Can you put that into context in terms of how significant this is?
F.U.: While it is large number in magnitude, it is certainly a small number compared to total emissions from Pfizer sites, which we do report annually in our corporate publications. Nevertheless, the accumulation of many small changes makes an impact, and is what we are committed to doing. To put this into context as you ask, based on EPA’s estimated average CO2 emission from a passenger car of 11,450 lbs/yr, it is the equivalent of removing 130 cars from the road.
PhM: How significant are the cost savings that you’ve realized from using less solvent and generating less waste?
F.U.: Very significant! Joseph Geiger of our Kalamazoo site provided me with some performance data, as an example. For the period inclusive of years 2006-2009, the Kalamazoo site recovered 21.7 MM gallons of solvents. The cumulative net savings (accounting for cost of recovery operations) in procurement and disposal of solvents is $65 Million! I don’t have data for our other operating sites, but they may be somewhat smaller due to smaller plant size, but I am sure they are in the tens of millions magnitude.
PhM: Finally, what does it mean to you to work with students on projects like these? Does it give you faith that the industry’s future in good hands?
F.U.: I’m proud that Pfizer sponsors such academic collaborations. The opportunity to consider solutions to real problems, particularly when there are broader societal or global implications, is a valuable extension of a rigorous academic program. It would be fantastic if all engineering students could benefit from projects like this, since our future thought leaders will come from this generation.
It was also personally rewarding to me to have worked with the students and the Rowan professors. It opened my eyes to the tools available for conducting sophisticated Life Cycle Analysis. It was also fun to have been able to introduce the students to the Pharmaceutical industry, to describe the regulatory requirements such as purity requirements and that validation would be needed to pool solvents from different products. However, there was a sad moment too, when I had to inform the team that project savings don’t all belong to Pfizer…taxes come into play in the ROI calculations.