Cultivating Contractor Relationships: Driving Value through Supplier Excellence
A few years ago, Wyeth Consumer Healthcare had a number of supplier quality issues that impacted key business metrics and compromised our customer service. Inconsistency and unreliability of supply cost us money, caused us downtime, and affected our ability to meet customer demands.
These issues were varied and for the most part unpredictable. They ranged from discolored or contaminated materials to malfunctioning components. What’s most important is that the potential for mis-orders was lowering our customer feedback ratings, and we were concerned about the impact this might have upon brand reputation as well as organizational credibility.
Our senior management said something needed to be done. So we established an effort to address these needs and prevent future supply quality concerns—the Supplier Quality Excellence (SQE) program. (Editor’s note: This article is derived from Ron Perry’s presentation from our recent webcast, “Operational Excellence for Building Better Partnerships”)
Foundation for Success
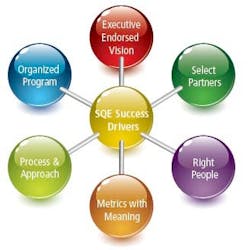
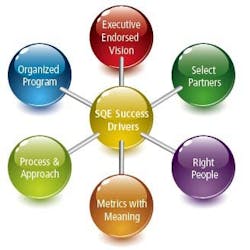
Our program success factors include an executive-endorsed vision, a very organized program, processes and approaches that are standardized, and metrics with meaning. We accentuate the right people, and we partner with the right providers (Figure 1).
Figure 1: SQE Program Success Factors
The Supplier Quality Excellence program works to improve suppliers who may be delivering poor and inconsistent quality. It works to increase existing supplier performance, whether it is poor, at par, or above par. And ultimately it works to try to drive greater overall value.
We’ve established partnerships with suppliers at each and every one of our manufacturing sites. We have carefully defined projects per each supplier and we try to employ standardized Lean, Six Sigma, and even Total Production System approaches to ultimately be the best. We aspire that one day people will point to Wyeth the way they do Toyota and say, “That’s the Wyeth Way.”
We have top-down executive management endorsement, and we believe it has been critical to our success. This program is a key strategic initiative within Wyeth Consumer Healthcare. It’s governed by a senior-level steering team which importantly includes representation from all our manufacturing sites. This is not just a corporate-run initiative. The engine of this program is embedded in each and every one of our sites that interact with suppliers and materials on a daily basis.
Our strategic initiative is defined beyond its vision. It has a specific mission: Reduce the risk of product supply interruptions due to raw material and packaging component issues. Develop a mutually beneficial relationship with key, selected suppliers to proactively prevent these occurrences.
And we have established goals, priorities, and key metrics. Our top-line metrics are very simple. They are about expanding our program and having a greater impact on our suppliers. It’s about reducing the potential for unwanted events and controlling a lower level of events. And it’s also about reducing the potential for a supplier event to occur. We expect the metrics of our program over the next few years to include more measures on total value, and more clear connections to budget reduction and operational efficiency, and whether we’re using our resources the best way we can.
The program started about two years ago with concepts. We had many non-standardized approaches, but we had people working within our North American and European sites that had great ideas, and they were trying things on. Our team members frequently shared their approaches and best practices. They decided to replicate what they felt worked best. And they ended up putting together standard practices that we use today and try to improve upon.
I’d like to note, citing author Jeffrey Liker, that standard doesn’t mean permanent or unchangeable. It does mean consistent and improvable.
Focus
Figure 2: Example of SQE Progress Monitoring
Critical to our process is making sure that each and every team that is working with a supplier on a particular project has focus. We have focus by using project charter documents to know what it is that we’re aiming for—the clear endpoints and success measures (Figure 2). We have the ability to monitor progress on our agreed-upon targets, and when they’re achieved. And we capture that history and manage that knowledge.
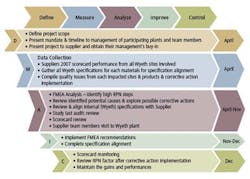
Our experienced green and black belts quickly apply the DMAIC approach (Figure 3). This is a standardized process we use in every single one of our programs. We have quality training, development, and experience, all of which has proven essential to the success of each SQE initiative.
Within the programs, we employ failure mode and effects analysis (FMEA), and this has become a tool which each supplier uses outside the program, even with their own supply chains and with other customers. This has been an excellent and comprehensive measure for assessing where the potential or likelihood of risk may occur. It’s fostering a Mutual Quality System and helps us to estimate the probability of issues.
Additionally, there is a softer side to the program. Stealing from the Toyota way, Genchi genbutsu, or “Go and see for yourself,” includes our engagement with all levels of suppliers—even shop-floor level. It’s personal. It’s high touch-point. You can apply it anywhere. The value here comes from observation and from learning about how each company can improve, recommend changes to one another, and improve the overall value chain.
Keeping Score
The SQE program isn’t just one-directional. The program includes a reverse scorecard which helps assess a supplier’s perspective on us. The feedback is essential, to be used as a mirror so we know how to change, where to focus, and what to prioritize. We use this with our own customers, called “Voice of the Customer,” so why not do it with our own supply channel?
Measures and metrics are important, but we have different kinds. We have what we call what metrics, which are the specific, outcome-based results of each program that we put in place.
But equally, if not more importantly, we have how metrics, which are relationship-based and about how we work. They’re about the behaviors that are being exemplified and how we’re changing behaviors and attitudes, and the way we work to construct relationships that are sustainable to drive greater value with suppliers. We believe it’s the behaviors that count and that become standard practices in the way we work.
It’s About People
We have a multitude of project-level metrics, but ultimately many of them are focused on quality issue reduction, performance improvement, and the prospect of quality assurance.
But it’s not just about the suppliers, the processes, the tools, the governments. It’s about the people. We created a special role at each of our manufacturing locations called the Supply Base Manager. The role of this person is to focus on the activities that we’re describing, ensuring that suppliers and their performance are managed effectively and working deeply with all functions within sites, above-sites [corporate], and certainly throughout a supply chain to serve in a preventative way to ensure quality, as well as in a restorative way.
Figure 3: The DMAIC Approach (click image to enlarge)
A Supply Base Manager:
- Ensures suppliers and their performance are managed effectively enabling site operational excellence
- Assures site commodity and category needs addressed through effective sourcing
- Supports Corporate and provides support for site-based supplier strategy & plans
- Evaluate current & select new suppliers and materials
- Serves as critical site-team player as new sources and materials are being introduced
Selecting Partners
Regarding partners, we have a selection process to understand where we should focus first. Our partnership and program selection are carefully planned and managed. We decide around purpose, we decide around value for both parties. We look at suppliers input as well as our own input.
We use several criteria to screen and select suppliers and projects (Figure 4). Ultimately, approvals are gained internal to the company and externally with our suppliers.
Figure 4: Supplier Focus Areas
Results
Results are looked at in several ways: recognition, reduction, relationship, and replication.
Recognition: This program was recognized by Pharmaceutical Manufacturing for its 2008 Team of the Year for large companies. Internal to Wyeth, we’ve been recognized by the president of Consumer Healthcare with his Global Pride award. We’ve also recently been recognized throughout our Wyeth network, across all divisions, as a best practice program with an [Network] excellence award. You can see the results that partially justify the recognition:
- 68% Reduction in Supplier Investigations
- 53% Reduction in potential supplier issue occurrences (RPN)
- 90% Supplier Scorecard use common performance scorecards.
- 80% of suppliers with Kaizen programs
- Structured communications with 80% of participating suppliers
- 60% Go-See “genchi genbutsu” visits
The rest of the justification comes from the hows and acknowledgement of the way we work. These are significant mile markers and progress towards being the best. Finally, recognition is also how our suppliers notice changes in their own facilities.
Reduction to us means measures to reduce cost, waste, cycle time, resources, inefficiencies, and transport. We have quantifiable reductions in all those areas, including energy use. Reduction in total costs and non-quality costs continues to be an area of focus for us because it goes far beyond just reducing quality incidents. It’s about operational excellence and efficiency in our organization. This is what is essential to us, not only to have a strong program today, but to have a sustainable program that brings value back to our bottom line tomorrow.
Replication is the core. This isn’t an isolated program. It has been picked up and stolen shamelessly across several biopharmaceutical, pharmaceutical and nutritional plants that we have across the Wyeth network. We’re very proud of this because people are pointing to us and trying new practices and techniques, and feeding back to us what is working so we can improve our program as well.
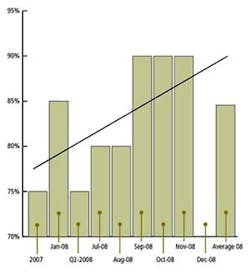
Figure 5: Documented Improvement in Service
Relationship: Importantly, it does relate to customer service. Here’s one example of how our customer service measures have increased out of one location from 75% to 90% throughout 2008 (Figure 5). How does it relate to our brand image? Customers are telling us they’re more satisfied. With greater confidence and better feedback, can you see how marketing can now make greater plans and commitments with new and existing products and markets? We can see that, and it gives us greater confidence and greater predictability to figure out how our top line will be affected.
This program for us is structured, is Lean-focused, and it’s driving efficiency, waste-reduction, and cost improvement. We have greater assurance of the customer needs that we serve, and also know that we can go to sleep at night not being fearful of inconsistent, unreliable, unpredictable quality, which are the concerns that I shared in the beginning.
We know that we can increase our confidence and business continuity, and our compliance. We’ve limited our liability. But we also recognize that there is always much room for improvement. Using the right people, partnering with the right suppliers, and working in the right ways in new and existing relationships are all critical to our success.
About the Author
Ron Perry leads a global sourcing team responsible for Wyeth Consumer Healthcare's (WCH) production materials and contract manufacturers, and is co-champion of WCH's Supplier Quality Excellence program. He has an MBA with emphasis in Finance & Operations from Penn State University, and earned his B.S. from Michigan State University.