Operational Excellence: Pharmas Missed Opportunities
Few managers of pharmaceutical companies see manufacturing as a competitive advantage today. Increasing cost pressures in the Industry have led more and more of them to take advantage of the global low cost sources for the production of goods while reducing their own production capacities.
Although, on surface this makes sense, this strategy could ultimately harm the competitiveness of pharmaceutical companies in the long term. In most cases the outsourcing decisions are based on a simple comparison between the costs to make a product at one’s own location and the costs in the target country!
Besides the issues that are going to be discussed below, separation of manufacturing from R&D has long-term consequences on the research and development process. Manufacturing input is essential for high quality product development and there is a need for constant dialogue between the manufacturing site and the researchers who are focused on just developing a laboratory process.
The Europe-wide benchmarking study conducted by the University of St. Gallen, Switzerland, and the International Association for Pharmaceutical Technology (APV), reveals that an average pharmaceutical plant could save up to € 6.5 million per year by achieving the degree of operational excellence equivalent to that achieved by the top 10% of the sample. About 100 pharmaceutical manufacturing locations all over Europe were analyzed in this study. Nevertheless, the challenges in grasping this potential are many fold, and many companies struggle to achieve a sustainable success. Through an analysis of the data from this study and from experiences gained through site visits in the European plants of a big research driven company one can derive the main success factors for the sustainable implementation of operational excellence.
Effectiveness Before Efficiency
Our data provides evidence that if an operational excellence initiative first tackles effectiveness issues and only thereafter shifts its efforts to increase efficiency; the program is much more likely to succeed than if it’s done in the reverse order. Unfortunately, this does not always reflect the interests of Senior Management in every company. As the big savings come with the efficiency driven parts of the programs, such as Lean Manufacturing, some of them are focusing on efficiency right from the beginning.
These programs are likely to fail in the long run, since at the end of the day, no sustainable success beyond some initial gains will be achievable.
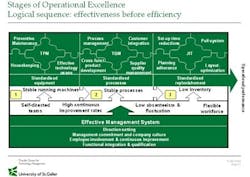
To achieve a high JIT Performance, a stable underlying system comprising the equipment and the processes in place is first required! A closer look at a sample revealed that the best performing companies in the Just-in-Time category (this category comprises KPI’s like inventory turnovers, throughput time, set-up time, etc.) have at the same time a significantly higher performance concerning Total Productive Maintenance (TPM) and Total Quality management (TQM) than the rest of the sample (Figure 1).
However this does not work in the reverse order. Being a TPM Performer does e.g. not help to predict a high JIT performance. Therefore a sustainable initiative must start with actions related to reduce the variability of the core manufacturing processes. For example, reducing the percentage of unplanned maintenance by doing more preventive maintenance could stabilize operation of the equipment as a first step. Learning more about the process and reducing variances (e.g. by applying Six Sigma) will help to increase the quality of the manufacturing processes. As soon as pharmaceutical manufacturing plants can ensure stable running equipment, stable running manufacturing processes and reliable and integrated suppliers, a JIT-program can help to reduce buffer stocks and increase the flexibility. An effective management system has to support the implementation of these steps in the right order:
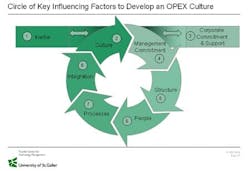
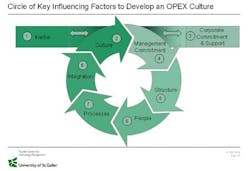
Success Factors in Striving for Operational Excellence
However the biggest challenge in improving Operational Excellence is to manage a paradigm change at the sites convincing people that they start to think in continuous improvement. This challenge is never really over. Therefore, Operational Excellence is a continuously ongoing journey that requires management attention on each level!
In the visits to plants all over Europe we could derive the following success factor supporting the sustainable implementation of Operational Excellence!
Show Corporate Commitment
There have been too many poorly designed restructuring programs in the past. So employees are not always sure how seriously a new program should be taken. Sometimes, their attitude is just to wait until it’s over again! Therefore there must be a noticeable commitment of corporate management to the excellence program. Too many companies have tried to manage their corporate excellence programs from behind the desk! What is needed is physical presence of responsible managers at the sites, where change has to happen. The responsible managers for operational excellence on the corporate level will have to be senior managers (often having managed a site by their own before) so that they can talk to the site managers face to face. Next building up operational excellence needs dedicated people dealing with this. Fostering Operational Excellence is not a part-time job. Corporate budgets will be mandatory for the implementation.
Understand the Program and its Link to Strategy
To get a buy-in on all levels in an operational excellence program it is mandatory that people understand the meaning and the objectives of the program. The objectives have to be linked to the overall strategy of the company and the site. This will ensure that the program is seen as improving the survival and the competitiveness of the plant and not simply as a “pain in the neck”!
Gain Management Commitment on Plant Level
The site manager faces tremendous pressure in today’s business landscape struggling with low cost competition in- and outside the company. Usually they have to deal with a lot of different initiatives at the same time such as PAT, QbD, Six Sigma, Lean Production, etc. In order to obtain management commitment, it is mandatory to explain and manage Operational Excellence as an umbrella initiative helping to structure all other things going on.
This will ensure that the site management understands that striving for Operational Excellence will remain as an objective even when one of the sub-initiatives takes a break. This understanding will help to get a site management commitment for Operational Excellence and give orientation to the whole plant. A common misunderstanding is e.g. to believe that Operational Excellence is something that starts in the plant! Preventive methods like QbD ensuring that a process is well designed from the beginning are also part of Operational Excellence.
Give Structure and Resources to the Program
In the beginning, there is a strong need for a support organization in each plant to help start Operational Excellence. Somebody at the plant has to be the coordinator of all activities related to improving operational excellence. He has to coach ongoing activities and has to ensure that there is a close link to the plant leadership team. Without a dedicated leader for operational excellence on the site level, the efforts will usually only have limited success.
Communicate
Communication to the shop floor about operational excellence is an important factor in getting a commitment of each single employee to continuous improvement. At the launch of the program, the communication will be about the basic idea, the business logic, the number of started improvement projects etc. As time goes by, the character of communication has to change. People will want to see the success of the activities, the gains achieved, and the improvements in their operations.
Select the Right People
Improvement projects in Operational Excellence usually require the working together of cross-functional teams. The challenge in achieving results is mostly not a methodological or a technical one but a management challenge. The key capability for successfully completing a let’s say 6 sigma black belt project is management! This means that one has to select people as team leaders, yellow, green or black belts that have management capabilities. Without this, one will not see any return from the projects. This means that one faces a certain trade-off as the same people are needed to run the day-to-day operations too.
Select the Right Projects
Quite often, while starting with an operational excellence program there is usually no clear selection process for improvement projects. In some companies it’s even worse: since they have e.g. put a mechanism in place that one can qualify as a green or black belt only when one has done one’s own project, a lot of projects are started with no clear business logic but only to have done it. Such a mechanism can again endanger the image of the operational excellence program in general. The conclusion is that it’s mandatory to define clear criteria based on business objectives a project has to fulfil to get started.
Control the Success of the Projects
It is an old saying that “what gets measured, gets done! Therefore what is needed is to establish some simple KPI’s showing that the program is running and is achieving results. KPI’s could be number of running projects, number of projects in the pipeline, cancelled projects, projects on hold, achieved savings, people trained,etc.
Ensure Knowhow Transfer
As Operational Excellence will be widely distributed in the overall production network of a pharmaceutical company it makes a lot of sense to establish a mechanism to ensure knowhow transfer between the sites. A good way to do it is to standardize the form of documentation of the projects and to make available the documentations using a centralized data base. As an additional measure, leading companies build up a network between the people responsible for OE from the different sites. They discuss with one another in periodical meetings and exchange ideas.
Don’t Forget People
Most important is like in many other change programs that you can not change when you’re not systematically involving people in this change. Most of the success factors discussed here, help to make people engaged in the required changes. One can not order continuous improvement from the top!
Summary
The Strive for Operational Excellence is a journey and leading companies see it as a never ending one! It needs a careful design and the willingness to overcome initial problems for a sustainable implementation of an operational excellence program. Figure 2 shows the most important dimensions that have to be taken into account climbing the mountain to excellence! In today’s business climate, that is not easy as Senior Management often go for the quick wins instead of investing in something sustainable. This also explains why more and more companies outsource their operations or let consultants do headcount reductions instead of increasing their manufacturing excellence.