The 2016 IFPAC conference venue was a combination of a “good news/bad news” statement: the good news was that its location placed it within a short drive/Metro ride from FDA’s headquarters in Maryland. The bad news? It was held in late January…and despite protests to the contrary, it always snows in the N. Virginia-DC area in January. And, my goodness, did it snow this year; nearly three feet, in some places, making travel, at best, difficult (a five-hour wait for a cab?). Despite the delayed and cancelled trains, planes, and automobiles, run, the conference did. And, based on some of the papers I attended, it was worth the effort.
SESSIONS
With sessions moved to new times and rooms, speakers absent, and workshops placed at strange times, it was a test to find and attend them. However, the challenge proved worth it. One nice example was Sanjay Sehgal’s (Aprecia Pharmaceuticals) talk about 3-D printing of their products. This was one of the best demonstrations of how the dosage forms are produced. The continuous line, where layer upon layer is applied.
Assembly of the megaPOD; top section, left, lifted and bottom section, left, being moved into place. (Courtesy of G-CON Mfg. Inc.)
Bogdan Kurtyka (USFDA, Silver Spring, MD) gave a number of good and specific examples of when, why, and how to perform life-cycle management on near-infrared procedures. Unless one has practical experience with NIR, university training and general knowledge of GMPs will not help. Kurtykaused charts and graphs to show when a change in the instrument will or will not trigger recalibration; when to routinely challenge an equation and how; what type or transfer is a major or minor change. Not the flashiest topic, but basic to successful application of NIR to PAT/QbD.Line Lundsberg-Nielson (Lundsberg Consulting, UK)explained the EMA’s 2014 Regulatory Guidance on NIR Spectroscopy. She showed how the EMA document compared with the recent FDA Guidance, the USP chapter, and ICH Guidances. Much like the FDA Guidance, it is strongly influenced by ICH Q2 (Validation of Analytical Methods) and attempts to bend parameters designed for HPLC to work for spectroscopic methods.
There are some minor misuses of words in theNIR Spectroscopy document (i.e. transmission/transmittance, reflection/reflectance) which are not significant, but the use of “correlation coefficient” for linearity is somewhat disturbing (addressed in my Therapeutic Dose column last year). In liquid chromatography, it is quite simple to make standards from 70-130% of label (or, as some do, 50-150%), since these are synthetic solutions. In a process analysis, where solid dosage forms are examined, it is seldom easy or advisable to attempt making tablets at these ranges (physically, they may not even be able to be made). The “linearity,” when based on correlation coefficient, may be influenced, not by improved accuracy, but by merely expanding the range of sample values, which muddies the waters for evaluating the worth of a NIR method.
I assume the difficulties will be addressed after comments are received by EMA and will be addressed in the next version. Otherwise, it was quite a nice presentation.
There were a large number of papers devoted to continuous manufacturing and Raman at this year’s conference. Both CM and non-typical monitoring methods are good signs and I look forward to hearing even more, next year.
EXHIBITION AREA
There were a goodly number of booths to find some nice equipment, but I want to highlight a few. There were a number of Raman instruments on display, but one stood out. Timegate Instruments' (Oulu, Finland) booth was worth a stop, for example. The pulsed LASER source is at a visible wavelength, generating a maximum Raman signal, but, as per theory, a huge fluorescence signal as well. However, in this instrument, the excitation of the sample is followed by an electronic “slamming of the door,” timed to admit the majority of Raman radiation, yet shutting out the interfering fluorescence radiation.
The sensitivity of the equipment answers the charge that Raman is not sensitive enough to measure trace components. While nowhere near “process-ready,” the instrument is, after all, only two years old. I can only imagine how the design and applications will progress in time (the design evolved at VTT, Finland, after all and they are known for their engineering acumen).
Advantest America was showing more applications of its TeraHertz instrumentation. They have moved from “look at the interesting toy” to “see what we can do in the process line.” They have shown their ability to monitor (and, with proper interfaces) control ribbon compactors in real time and appear to have a good handle on tablet coating, both in-process and post-process.
Umetricsis now known as MKS Data Analytics Solutions. Still offering their SIMCA Chemometric/statistical software and Design of Experiments package, they are expanding to on-line process monitoring and control software. Anyone interested should check out their webpage for details. But, based on the quality of their earlier work, I suspect this endeavor will be worth watching.
Microtrac was offering some interesting particle characterization equipment. I was impressed with the multiple LASER diffraction units (various colors and up to three LASERs). Of course, they offer Zeta potential, 3-D imaging, molecular weight and surface analysis devices. Being over 40 years old, they seem to have quite a bit of experience in this field.
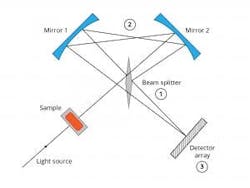
Keit (Oxfordshire, UK) was showing a rather interesting FTIR, theirmicroFTS, originally designed for use in satellite, is low weight, compact, and extremely rugged. Unlike most FTIRs, it uses a beamsplitter (1) to direct light in both directions around a loop interferometer (2) that contains no moving parts (see below). The whole interferogram is formed on a detector array (3) at the same instant. This configuration means that noise or vibration affects both beams equally and is (largely) cancelled out, making the data exceptionally robust. This rapid data generation may be a perfect answer to problems with moving samples or rapid reactions.CAMO Software AS (Oslo, Norway) was there, as usual. I didn’t have a chance to play with their latest edition of their Unscrambler software. However, if it is as good as all the other previous versions, it is well worth the time to investigate. I have had excellent success with Unscrambler over the years and their customer service is second to none.
BrightSpec (Charlottesville, VA) was a brand new company at last year’s conference. In case you don’t have an archive of my columns, the instrument is, in effect, a vapor-phase NMR (Fourier Transform Molecular Rotational Resonance Spectroscopy, to be more specific). FT-MRR spectroscopy is a novel technique for, for instance, fast genotoxic impurity and residual solvent analysis. The FT-MRR impurity analyzer can monitor a broad range of compounds with the particular subset chosen and modified by the user. Both static headspace methods at the bench-top and direct analysis of vapors in drying chambers are possible.The headspace or off-gassing vapors are introduced into the spectrometer cell and each analyte of interest can be quantified in seconds. FT-MRR spectroscopy can be used to determine both simultaneously.
And, I am told, several of the instruments now are being used in industry and that we should expect a spate of research/apps papers in the near future.
GEA Pharma Systems (Wommelgem, Belgium) was mentioned by a number of speakers and other with whom I conversed, I thought it would be worth adding to the conversation. GEA provides the hardware and systems for continuous manufacturing (see my recent write-up for this magazine for more information on CM) for a number of large Pharma companies. GEA recently announced a collaboration with GSK to further develop self-contained, POD-based mini-factories to develop and manufacture pharmaceutical oral solid dosage (OSD) forms. This partnership expands on GEA’s existing alliance with Pfizer and G-CON Manufacturing (see the article about this published in June 2015 issue) to design a portable, continuous, miniature, and modular (PCMM) prototype unit, which is currently being implemented at Pfizer’s labs in Groton (Connecticut, USA).
Conceived in conjunction with Pfizer and G-CON, the three companies formed a consortium to design and build an autonomous manufacturing environment for continuous OSD production using GEA’s ConsiGma 25 equipment and G-CON’s modular POD system. Sounds like the future is closer than it appears (in your rear-view mirror).
NETWORKING
The best information, it turned out, was gleaned at the bar/restaurant next to the lobby. Conveniently placed between the entrance and elevators, one could see anyone of interest and have a conversation with them. Since the weather precluded outside trips, I had a captive sampling of researchers, both industrial scientists and instrument vendors.While I never “kiss and tell,” many of the conversations I had will appear in columns, later this year.
All in all, while a little confusing, somewhat truncated, and isolated, the 2016 IFPAC meeting was well worth attending. Hopefully, Mr. Zudkis will listen to the many voices suggesting swapping the dates of the North American and Puerto Rican meetings; January in Puerto Rico is nicer and considered “off-season,” so hotels shouldn’t be expensive. And, Virginia is quite pleasant in June. Anyone second the motion?