Rutgers Engineering Center for Structured Organic Particulate Systems (C-SOPS center) in New Brunswick recently opened its doors to Pharmaceutical Manufacturing for a first-hand look at its CM research program. The result of a close collaboration with Janssen and a number of academic, public and private entities, according to Rutgers “C-SOPS brings together a cross-disciplinary team of engineers and scientists, as well as industry leaders, to improve the way pharmaceuticals, foods and agricultural products are manufactured. C-SOPS will focus on advancing the scientific foundation for the optimal design of SOPS with advanced functionality while developing the methodologies for their active control and manufacturing.”
The overall objective of the project is to develop how to use raw material properties, formulation composition and process parameters to predict the properties of intermediate blends and finished direct compression products made in the existing INSPIRE (RU-CM) line.
RENEWED FUNDING
Figure 1. Excipient/API hoppers and weighing units occupy the top tier.
In May 2015, Janssen Supply Chain announced it had expanded its collaboration with Rutgers and the School of Engineering with a $6 million funding arrangement to help create the future of advanced manufacturing support for the school’s Continuous Manufacturing Initiative. The funds from Janssen, part of the Janssen Pharmaceutical Companies of Johnson & Johnson, will increase research and development efforts at Rutgers’ C-SOPS over the next several years. The center is helping transition several products to continuous manufacturing, including developing a specially designed manufacturing line at Janssen’s new facility in Puerto Rico.“We are very pleased to broaden our collaboration with Janssen on this important project and look forward to expanding collaborations with other pharmaceutical companies globally,” said C-SOPS director Fernando J. Muzzio. “Continuous manufacturing represents the future of advanced pharmaceutical manufacturing and will produce significant improvements in efficiency, safety, cost and speed to market. Our center is now better positioned than ever to serve the scientific community, the pharmaceutical industry and the regulatory agencies.”
According to Janssen, Rutgers established the engineering research center nine years ago with funding from the National Science Foundation and a consortium of more than 40 companies within the pharmaceutical sector, and is the world’s largest academic-based research organization dedicated to modernizing pharmaceutical manufacturing of solid oral dose medications.
The funding from Janssen represents a significant expansion of an arrangement that began five years ago and that focuses on technical development of continuous processing, an emerging advanced pharmaceutical manufacturing method. Besides the investment from Janssen, Rutgers anticipates another $1 million from the U.S. government as part of an ongoing FDA initiative. Anticipated outcomes include:
In-depth knowledge of the effects of material properties on product performance for existing CDC line, and General methodology for developing “formulation space models” using the available CM (continuous manufacturing) line as a first scenario, but implemented in a manner to facilitate the extension of the methodology to other continuous manufacturing systems.
In other words, the Rutgers’ team is attempting to build an ab initio approach to continuous manufacturing and real-time release (RTR), replacing the current ad hoc (product-by-product) approach.
To accomplish this end, a series of technologies are used, both in the lab and online, to characterize the interactions of raw materials and intermediates and how these affect both the process and finished product behavior (Critical Quality Attributes). The studies are designed to both characterize materials and give tools to developers with which to use these data to speed their own designs.
Figure 2. In-line ribbon compaction device.
One example of a lab-based instrument is simply a rotating, clear cylinder with sensors located below and a series of light sources at one end. The device then has a known amount of powder or granular material added and is rotated along its long axis at various speeds. The combination of visual observations and weight distribution allows the researchers to characterize the flow (or sticking/lack of flow) character of any material.These data are combined with the results of the Powder Rheology device. This rheology is, in essence, the “viscosity” of powders and, when combined with the “flowability” results, gives a clearer picture to formulators as to what to expect in a process stream. It allows them to anticipate the need and level of slip/flow agents for a particular formulation, without spending days or weeks experimenting with actual runs. Some of the production steps where this information may be used are:
Blending APIs and excipients. One problem in most blenders is the potential for “dead spots” or areas where material flow is stagnant, causing areas on non-mixed materials. These data could assist formulators in both choosing the proper blender and knowing whether a lubricant is needed and, if so, which one and how much is needed.
Tablet coating. Since most tablets are still coated in a rotating tablet pan, the sliding of tablet-over-tablet throughout the coating process is critical to an even film on each tablet. These tools could be used to assist formulators in designing the proper methodology.
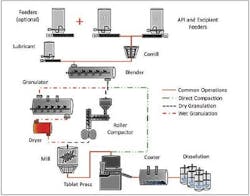
Before addressing individual components, a look at a few schematics of the overall process with alternate units for varying granulation approaches may be enlightening. Figure 3 shows a CM set-up that can either use conventional granulations or roller compaction. Many formulators, using CM units, are eschewing wet granulations because of the drying times. The process unit is one of the more complicated and expensive parts of a CM process.
Figure 3. A CM unit with both a roller compactor and Granulator. The set-up allows either to be used, depending on which is required.
A second CM process reveals the use of direct compression in lieu of any granulation step. If this is possible for a product then several steps (granulating, drying, particle sizing, lubricating) can be avoided, speeding up and simplifying the process. This aspect of process emphasizes the need for tight computer control of each step with transparent feed-back and feed-forward options. That is the heart of QbD (Quality by Design), wherein the operators are allowed (and encouraged) to modify operating parameters throughout the production cycle.Examining the individual components shows how interchangeable and easily scaled (up or down) they are. The “swap-ability” of the individual components, coupled with the ability to change operating parameters, avoids the necessity of revalidating the process each time it is modified.
A weighing platform on the top tier of the prototype unit at Rutgers is shown in Figure 4. The hoppers may easily be expanded and more added, as the formulation requires. The powders (APIs/excipients) are dispensed by weight, making the physical parameters (i.e., density, flowability, particle size distribution) important for correct dispensing.
Since tableting and encapsulating machines operate on volume, not weight, it is critical that the correlation between weight and volume (e.g., density) be monitored and accounted for throughout the entire process train. This is where the body of knowledge that the Rutgers’ team is building; having an idea of the physical characteristics of raw materials allows formulators to both formulate and produce better products.
Figure 4. Screw-type powder blending is accomplished on the second tier. Tableting and coating steps occur on the third, lowest tier.
An Avogadro-screw type mixer on the second tier (Figure 1) anchors a process where powders enter one end of the mixing tube, are continuously mixed (screw-action), and with the proper mixture emerge from the far end. Again, this unit may be replaced or upgraded, but, using a properly constructed Design Space, revalidation is not necessary. Monitored with NIR or Raman sensors, the homogeneity is controlled such that, if it does not meet pre-determined specifications, the process either stops or the mixing modified (screw speed, ingredient add speed).The continuous stream granulator is one of several commercially available and, as with the other units in the CM train, can be upgraded or replaced with impunity, since a proper design space will allow operators or formulators to adjust to any mechanical differences without the need to revalidate.
ALTERNATIVE TO WET
An alternative to “wet” granulation (and subsequent drying and sizing) is seen in Figure 2. The ribbon compactor “squeezes” the mixed powder into a continuous, semi-elastic ribbon. If performed properly on a well-blended mixture, the resultant ribbon, when chopped to granules, gives an equivalent to the granulation product. It is then lubricated (with e.g., magnesium stearate) and transferred to the tableting or encapsulating machine.
The tableting machine (RU unit) is shown in Figure 4. As with all the other units in the process stream, the tablet press can be replaced or the powder stream split into a second unit. Thus, the production rate may be increased by enlarging the press or using two units. The feed from the press may then be shunted into a continuous coater (and dryer).
OBSERVATIONS/COMMENTARY
The overall purpose of the cooperative program at Rutgers (and other universities) is to take the years of knowledge about tablet and capsule formulation and production and systematize the information into cogent rules and guidelines for future formulation work. As mentioned previously, formulating one product at a time is outdated; for the industry to embrace continuous manufacturing, this type of work is critical and hopefully will lead to guidelines and more theoretical foundations to build on.