Exploring the Benefits of Real-time Particle Sizing
The optimization and scale-up of wet granulation processes is often an essential step in the commercialization of oral solid dosage (OSD) products. Granulation enables the preparation of an optimized blend for tableting in which small quantities of highly potent active ingredients (APIs) are stably and homogeneously distributed. Furthermore, granule properties can be manipulated to control, for example, the flowability of the blend and its compressibility. In these ways granulation underpins the manufacture of high-quality tablets with consistent dose uniformity.
The technique of spatial particle velocimetry (SPV) has been used to develop PAT, exemplified by the Parsum inline particle sizing probe, which is well-suited to high shear granulation applications.
Particles falling through a laser beam cast a shadow, interrupting the flow of light to a linear detector array. The sequential interruption of neighbouring elements of the spatial filter detector triggers a burst signal, the frequency of which is directly proportional to particle velocity. A secondary signal called the pulse signal is also generated. This is a measure of the length of time for which the particle blocks a single detector. In combination, the burst and pulse signal allow the determination of a particle size based on the chord length of individual particles. These measurements are used to develop a number-based particle size distribution.
With SPV, measurement rates are sufficiently fast for real-time process monitoring. Parsum SPV inline probes have no moving parts, measure across a range of 50 μm – 6 mm, and are available in sizes suitable for measurement from lab to pilot and full scale, to support process scale-up. These instruments work reliably, even at the high and variable particle loadings encountered in a high shear granulator, providing an appropriate dispersion system is in place. Continuous purging of the optics ensures robust measurement even within hot, sticky, fouling environments.
APPLYING HIGH SHEAR GRANULATION
The first step in producing optimized granules for tableting is the wet granulation itself which produces agglomerates that range from a few hundred microns to several millimeters in diameter (see Figure 1). These are typically wet milled (de-lumping) prior to drying in a fluidized bed dryer. Subsequent milling of the dried particles produces a closely specified granular feed for the tablet press.
Figure 1: Well-controlled wet granulation produces optimized granules for tablet production.
Focusing on the wet granulation step, this can be further broken down into a number of stages. The first, pre-mixing, involves dry mixing of the components of the blend where the API is often present as a micronized powder and excipients are in the form of powders with a larger (typically 50-200um) average size. Once pre-mixing is complete, granulation liquid is sprayed into the mixer to promote cohesion of the powder particles. This liquid may be either water or a solution containing a dissolved binder. A second stage of liquid addition promotes further granule growth, to an acceptable finished granule size, as the mass is mixed to completion of the granulation process.The performance of the high shear granulation process and the properties of the resulting granules, are controlled by the following variables:
• The composition of the blend;
• The amount of binder liquid added;
• The rate at which the granulation liquid is added;
• The design and operating conditions of the granulator such as speed of rotation of the mixing blade, and
• Mixing time.
When applying QbD, the aim is to control the manufacturing process to ensure that the critical quality attributes (CQAs) of the pharmaceutical product are consistently met. Typically the CQAs for tablets are properties such as hardness, dissolution profile and content uniformity. Fully scoping the design space for the wet granulation step therefore involves investigating how the processing variables listed above change the trajectory of the granulation, its outcome — the properties of the finished granules — and ultimately the CQAs of completed tablets. Continuous particle size analysis allows real-time tracking of the particle size of the granules during processing, and therefore has the potential to accelerate such studies. The following case study demonstrates its application.
USING INLINE PARTICLE SIZING IN GRANULATION SCALE-UP STUDIES
A sequence of experiments was carried out to:
• Investigate the dynamics of wet granulation using real-time particle sizing and confirm the validity of linear process scale-up for a wet granulation process.
• Measure and compare the reproducibility of the granulation process at three different scales.
• Show that granules identified as closely similar, using particle size data, produce tablets of closely similar quality, as quantified by dissolution profile.
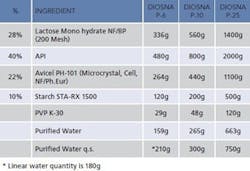
Table 1: Blend ingredients used to make granules for tableting.
Table 1 shows the constituents of blends for the comparative trial to investigate granulation performance on three different scales. A primary aim of this first step of the study was to confirm that the granulation process proceeded in an identical manner when linearly scaled. This confirmation would allow equivalent experimentation on either a smaller or larger scale, depending on other requirements of the research program, and directly supports process scale-up. An important point to note is that while in many instances determining the end point of a granulation is the primary goal, here the aim was to monitor the progress of the granulation over time. The focus of the study was therefore to demonstrate an effective method for monitoring and comparing the trajectory of the process.The tests were carried out using: 6 liters (Diosna P-6); 10 liters (Diosna P-10) and 25 liters (Diosna P-25) granulators. The relative proportions of each dry ingredient in the granulator feed, a mixture of excipients and API, were identical in each case. The total amount of material used was linearly scaled with reference to the increasing volume of the granulators, with the exception of the amount of purified water used for the smallest 6-liter scale unit. At this scale, linear scaling down from the amount of water used at the 10-liter scale gave a quantity that was insufficient to achieve successful granulation so a larger quantity of water was required.
Table 2: Granulation conditions applied during the production of granules for tableting.
The granulation conditions applied are shown in Table 2. The water was added during two granulation stages (as described above): Granulation I (PVP solution) and Granulation II (purified water q.s.). The pre-mix and spraying times were kept constant at 180 s and 130 s, respectively, at each scale. Linear scaling of the amount of granulation liquid added therefore resulted in an increase in spray rate as the size of the granulator increased.The Diosna granulators used in this study all have similar geometries and two integrated mixing devices: a centrally mounted mixer and a chopper installed within the wall of the granulator bowl. The speed of the mixer and the chopper can be independently set at either I or II. In Table 2 the speed setting for the two mixing devices is shown as being identical for each granulator, in each of the three stages of the granulation process. In practical terms this means that the tip speed of the mixing devices, which determines the power input to the granulator, was kept constant across the scales.During each granulation particle size was tracked in real-time using an inline particle sizing probe (Parsum IPP-70Se, Malvern Instruments). Experiments were conducted using two different mixing times, 300 s and 540s, to investigate the risk of over-granulation.TRACKING THE GRANULATION PROCESS
Scrutiny of the real-time particle size data generated during a granulation provides insight into how the granulation processes proceed. Figure 2 shows an example particle size trend from a single experimentation. The particle size parameters reported are x90, x50 and x10 which are the particle sizes below which 90, 50 and 10 percent of the particle population lies, respectively. Different stages of the granulation process are clearly observable.
These results show that, as expected, particle size remains constant during the pre-mixing stage. Neither does particle size increase substantially during the Granulation I stage, although there is a small rise in x90. This is attributable to the formation of a limited number of relatively large agglomerates which are subsequently broken down by continued mixing. In the early part of the Granulation II stage, particle size also remains constant but then, once spraying is complete, granules start to form, as mixing continues. Particle size rises quickly to a plateau. Even though mixing continues for some time after this plateau is reached, there is no further increase in any of the three recorded particle size parameters. This suggests that the process is robust with respect to the risk of over-granulation, an important finding.
ASSESSING THE IMPACT OF PROBE POSITION
To optimize the installation of the probe within the granulator, tests were carried out to determine any sensitivity to the positioning of the probe. The geometry of the P-6 and P-10 granulators allows the probe to be installed either at a 45° angle, facing the bowl wall, or at a 90° angle, facing the inside of the bowl. With the P-25 unit only the 90° configuration is feasible. Granulations were carried out with the same blend (see Table 1), and under the same conditions as described above (see Table 2) to confirm the reproducibility of particle size measurement.
With the smaller scale unit (P-6) the probe position has a significant effect on the reported size data, but with the larger unit (P-10) the results are insensitive to probe orientation. This suggests that probe position may be an important factor with respect to reproducibility and should be fixed to ensure relevant data for comparative, scale-up studies especially where smaller scale vessels are used. In vessels larger than 50L, orientation of the probe is guided by the general direction of the granulate flow and swirling effects induced by proximity to the chopper blade. An optimal position would allow the probe to meet the granule stream at a 45° angle to the disperser orifice.
ASSESSING THE REPRODUCIBILITY OF GRANULATION AND SCALABILITY
Further studies of reproducibility were implemented by carrying out multiple, identical granulations at all three scales. The aim of this part of the study was to carry out a rigorous assessment of reproducibility in order to reliably assess the scalability of the process. The composite results from these experiments, and those assessing the impact of probe position, are summarized in Figure 3.
Figure 3: Particle size measurements confirm that there is good scalability across the differently sized granulators.
Reproducibility is lowest for the P-6 granulator. At this scale reproducibility is impacted by the inclusion of results obtained at different probe positions, but even so there is no statistically significant variation in the results obtained. Granulation with the P-25 gives the most reproducible data: x10, x50 and x90 all have a RSD of less than 5 percent. Most critically, however, the results confirm good scalability across the three granulators. Linear process scale up to P-10 and P-25, and a constant mixer/chopper tip speed produces granules that are closely similar in size.ASSESSING THE IMPACT OF GRANULATION SCALE ON TABLET QUALITY
The results above show that each of the granulators produces granules that are equivalently sized, according to real-time particle size data measured in situ. They do not, however, confirm that each granulator produces granules that process equivalently in the tablet press to produce tablets with identical performance. This final stage of the experimental study was designed to assess whether differences in the granules emerge in the downstream processing steps that they are subjected to.
Samples from granulations at each scale were dried and milled. Particle size distribution data from the dried granules were then measured using a laser diffraction particle size analyzer (Mastersizer 2000, Malvern Instruments). Each sample of dried granules has the same particle size (data not shown), within the limits of statistical relevance.
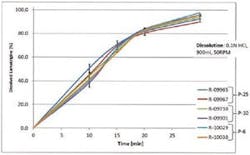
Finished tablets were then manufactured from dried samples from two batches, at each scale. Figure 4 shows the dissolution profiles of these tablets, a critical quality attribute of the product. The similarity of these profiles confirms that the wet granules from each granulator go on to produce tablets of directly comparable quality. This directly confirms the scalability of the granulation and that relevance of the data provided by the inline particle sizing analyzer for tracking the granulation process.
Figure 4: Comparable dissolution profiles confirm the scalability of the high shear granulation process across the 6 to 10 to 25 liter scales.
High shear granulation processes are a valued precursor to tableting, that help to efficiently deliver tablets with consistent dose uniformity. Appropriate PAT has an important role to play in helping researchers scope the design space of these processes, to mitigate the risk associated with the scale-up to commercial production. The results presented here show how real-time particle sizing in the form of an inline particle sizing probe can be used to successfully track high shear granulation. The resulting data speed up process development and enable the successful scale-up of this critical unit operation while reducing the numbers of trial runs and saving highly valuable API.