While biopharma’s parenterals may be the belle of the ball lately, Oral Solid Dose’s (OSD) dance card remains booked — the form of choice for thousands of medicines and billions of patients. Far from the dowdy, aging wall flower one might expect from such a mature segment, in reality some of the most exciting technological advancements in Pharma processing are occurring right now as Branded, Generic and CMO players reach out for more speed, more capacity and quality in solid dose cGMP operations.
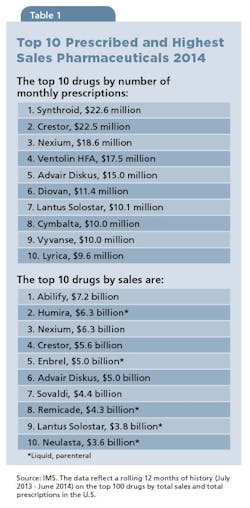
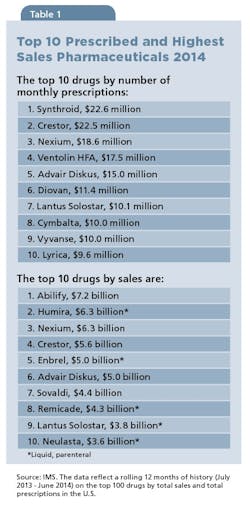
It’s always worth a moment to take a quick look at the category as a whole, just to calibrate on how important the OSD form is to the Pharma universe. With the exception of one product (Lantus), OSDs represent most prescribed medications. According to IMS Health, Synthroid (levothyroxine), with 22.6 million prescriptions, remains the nation’s most-prescribed drug; the antipsychotic Abilify (aripiprazole) at $7.2 billion, has the highest sales (see Table 1). Of the top 10 highest-sales drugs, OSDs lead, garnering $28.5 billion in sales versus $23 billion for liquid injectables.
There’s no doubt that contract manufacturing organizations (CMOs) are capitalizing on the continued expansion of the OSD market. In 2014, says Kate Hammeke, director of marketing intelligence at That’s Nice, the contract manufacturing market for solid dosage forms is projected to be $19.6 billion, representing 58 percent of the total CMO market, valued at $33.7 billion.
“While the market value percentage for solid dose has been drifting downward,” says Hammeke, “and likely related to the shift towards biologics — which are more expensive to develop and manufacture — the propensity to outsource oral solid dosage forms continues to grow modestly.” Nice Insight’s annual survey results indicated that solid dose manufacturing will be outsourced with the greatest frequency (55 percent) followed by injectables (50 percent), semi-solids (44 percent), then specialty dosage forms (42 percent).
Who’s outsourcing solid dosage forms? According to Nice Insights, Big Pharma is (60 percent) followed by Biologics (50 percent), Emerging Biotech (70 percent), Emerging Pharma (49 percent) and lastly Specialty Pharma (51 percent). Who’s getting the business? Some of the biggest names head the list (See Table 2), many offering margin-enhancing efficiency and expertise to those Big Pharma players looking to get the most bang for their buck out of patent-expired formulations.
Hammeke says those responding reported they would outsource finished dosage forms with a greater frequency than API manufacturing (for both large and small molecule APIs). “When it comes to outsourcing behaviors,” explains Hammeke, “respondents who will contract solid dose manufacturing in 2014 showed a greater likelihood for considering emerging market providers than the general population, with nearly nine out of 10 stating they include CMOs in emerging markets on their shortlists.”
She noted that, among cautions expressed by respondents to the Nice Insight study, intellectual property (IP) concerns were a top reason for not considering emerging market providers because this issue corresponds much more strongly to primary manufacturing/API production than secondary manufacturing of dosage forms.
Among the product types, controlled release tablets were of most interest to Nice Insight survey respondents (62 percent), followed by oral disintegrating tablets (56 percent), immediate release filled capsules (55 percent), and immediate release tablets (54 percent). Other types like controlled release capsules, powder-filled capsules and resin or bead-filled capsules followed at 49, 44 and 33 percent, respectively. Study respondents indicated that timed-release technologies, either rapid or delayed were of significant interest, but of most interest were technologies that protected formulations from stomach acids.
“Both outsourcing and offshoring have shown their efficacy in cutting costs for pharma companies when it comes to solid dose manufacturing,” says Hammeke. Another potential development to emerge from reducing capital outlay on in-house manufacturing equipment and technologies, says Hammeke, is the shift from tactical relationships for OSD projects toward more strategic, long-term agreements with manufacturers.
REACHING FOR MORE
Everyday, the economics of drug manufacture are growing more complex to manage effectively. If there is one thing for certain, the future health of the industry is resting squarely on the shoulders of its manufacturing operations, and clearly drug makers are striving to get the most out of their production capacity. Drug safety, supply, access, compliance and shareholder return all hinge on getting manufacturing and process operations right for both drug maker and drug owner. Industry behavior reflects how important manufacturing excellence is to business success and is reflected in recent merger and acquisition activity, the development of strategic and long-term manufacturing related partnerships, and strategic investment in new-era manufacturing technologies.
Without being too remedial, the traditional manufacturing oral solid dosage forms is a primarily a physical (not chemical) multi-stage process. This well-established process involves dispensing, milling or sizing particles and blending the formulation’s primary ingredients (APIs, excipients). Granulation is another primary step, and both wet granulation and dry granulation are used by manufacturers to prepare the formulation for direct compression into tablets or to fill capsules. Because a variety of processes can be applied to manufacture OSDs, including drying, compaction and coating, there are a number of factors associated with these fundamentals processes that can significantly impact the chosen form’s uniformity, stability and bioavailability.
REACHING FOR BETTER DATA
Benoit Igne, of Duquesne University (Pittsburgh), discussed in a recent PharmaManufacturing.com spectroscopy webinar, how closer control of the tableting process demands a better understanding of critical process parameters and critical quality attributes. For example, research conducted with Daichi Sankyo Co. at the university determined that one of the critical process parameters included pan rotation speed (the wrong speed could lead to erosion, breakage or winning), batch size, inlet and exhaust air temperature, inlet air volume, spray rate and time. Researchers monitored the coating and drying endpoint temperature, and applied Near Infrared Spectroscopy (NIR) in situ within the pan coater to estimate weight gain, moisture and tablet coater temperature.
Possibly the most common reason that companies fail at generating a Quality by Design (QbD)-based NDA or ANDA for OSDs is that the wrong measurements are “taken at the wrong place at the wrong time,” notes contributor Emil Ciurzak, Doramaxx Consulting. “All too often, a given Pharma producer’s ‘QbD team’ depends on classic techniques to build a Design Space,” says Ciurzak. “That means those folks may try to determine blend uniformity by stopping the blender and using a sample thief to gather samples or checking content uniformity by assaying 20 tablets with a HPLC.” To design good tablet-based drug-delivery systems, notes Ciurzak, manufacturers need to gain a better understanding of what happens in the making of tablets not after-the-fact assays. Says Ciurzak: “Quality by Testing is not QbD.”
Because the process of making a tablet is, as mentioned, a physical exercise and not a chemical one, after-the-fact tests like HPLC, hardness, friability and dissolution tell Pharma processors little about how the powder blends, flows, granulates, compresses and is coated. To obtain these data, says Ciurzak, in-process monitors are required to provide real insight into the “living process” of solid dosage form manufacture. “This is where PAT comes into the picture,” explains Ciurzak, “before a product goes into production, PAT gives us knowledge and a tool to control the process/product.”
Blend uniformity is a legal requirement, and Generic Pharma must, by law, test every batch for uniformity. Ciurzak notes the industry has slowly accepted NIR as a “reasonable” test of uniformity. More than a decade ago Pfizer and Zeiss presented the industry with the first wireless, portable NIR instrument. Now several companies offer these devices and the technology has been accepted by the FDA in NDAs and ANDAs. “The development of blend uniformity by wireless NIR was performed on product with 5-50 percent API content,” says Ciurzak, noting, “these levels are also quite within the wheelhouse of Raman instruments and have been the standard range of APIs for decades.”
LIF FOR LOW LEVEL API
With more potent drugs being developed and tested, the API level in the mix is often below 1 percent, making NIR or Raman detection (in a moving bed) problematic. A better technology for low-level API concentrations is Light-Induced Fluorescence (LIF). Until recently, LIF units had only a single wavelength operational range and induced most organic molecules fluoresce (to varying degrees). There were some specificity questions that slowed its acceptance. Tunable, multiple wavelengths and wireless operation are now available with newer units. These developments allow the formulator/operator to tune the incident light to the excitation wavelength of a low-level API, a lubricant or other organic ingredients in the mix. “Because not all ingredients mix at the same speed, notes Ciurzak, “this specificity allows any or all components to be monitored for completeness of blend.”
Powder Rheometers provide another way to measuring powder characteristics after mixing. The Pharma industry has been testing liquid viscosities for decades with similar devices, but now the technology exists where line managers can test the interactions between particles in a powder mixture. There are systems available now that combine shear, bulk, dynamic and axial powder testing methodologies for comprehensive powder characterization. Such information can deliver insight into parameters including flowability, compressibility and correlated to dissolution characteristics.
TO THE CORE
For some OSD manufacturers, testing tablet cores prior to coating may be prudent to explore and understand the characteristics of a number of critical parameters including: hardness, friability and tensile strength for example. Ultrasonic devices are now available to non-destructively measure parameters like the tensile strength of a finished tablet. The speed with which the sound travels through the tablet is measured and correlated with standard (destructive) testing technologies and the curve generated can be used to analyze process samples more rapidly. These data can be used to quickly predict physical parameters and allow formulators to understand what component ratio changes or compression changes are doing to the finished dosage form.
According to Ciurzak, unless direct compression, ribbon compaction, or extrusion techniques are employed, the most likely method of preparing the powder blend for compression is wet granulation. “Despite what some may think,” Ciurzak says, “wet granulation is not just a matter of pouring a given amount of liquid into a vat of pre-blended powders. Liquid needs to be added in a specific manner, at a specific rate, and usually from a specific position in the blender. Knowing what the manner, rate and positions are is the trick to a successful granulation.”
One way of measuring how a liquid disperses through a medium (powdered or another liquid) is tomography. A series of transducers is placed around a mixing chamber (glass or metal); by measuring such things as dielectric constant changes (varying permittivity), the differences throughout a mix can be visualized in real time. The multiple transducers deployed by these systems allow a 3-D picture to be displayed in real time. Using these data, mixing speeds, fill levels, blade type, addition speeds and points of addition may be optimized.
Coatings provide a number of positive attributes including stability, swallowability and cosmetic branding and identity. Coatings are also more technical in nature — now used to protect APIs and the physical make-up of the core, affect or control dissolution times and contain an active ingredient outside the core — thus they need to be accurately measured and controlled. The industry’s been using NIR in the coating pan for years to measure the amount of coatings and loss of solvents. In recent years, Pharma has seen Raman probes added to the coating control toolbox. The data generated from NIR and Raman probes has been used to determine a number of things including spray radius, solution addition speed, drying times, pan rotation speeds and exhaust functionality.
For understanding individual coating integrity, TeraHertz spectroscopy technology is a method worth exploring. Formerly known as “far infrared,” TeraHertz has been available for years and Ciurzak notes it may be used as simple spectroscopy but, in his opinion, its strength is in measuring interfaces between layers.
Of course there are numerous tools for fine-tuning dosage forms as part of a successful QbD program for OSDs. These and other technologies are maturing and becoming even more competitively priced and worth careful appraisal.
If you ask Sarang Oka and Fernando Muzzio, from the Department of Chemical and Biochemical Engineering at Rutgers University, the advantages of continuous manufacturing over batch manufacturing are well established. When properly implemented, they say, continuous processes are almost completely steady, can be designed at scale, and can be used reliably to minimize segregation and agglomeration of ingredients. Oka and Muzzio also maintain that continuous process is also the perfect platform for implementing PAT methods because the technology is necessary to ensure closed-loop control of continuous processes.
Continuous manufacturing’s business case is equally strong. In development, continuous manufacturing systems allow OSD manufacturers to perform complex DOE matrices in a few days using a tiny fraction of the material normally required to perform comparable studies in batch mode, enabling significant savings in labor, analytical costs and capital outlays. In his company blog, Emerson’s Jonathan Lustri explains that in light of all the market and competitive forces at work, in order for the industry to maintain profitability it has to increase its manufacturing excellence. For many OSD manufacturers, the best route to Pharma’s manufacturing renaissance is on the continuous manufacturing road. “The financial driver for the move to continuous manufacturing,” says Lustri, “is the significant reduction in capital and operating costs.” Rule of thumb metrics put typical batch process capacity utilization at about 35 percent. On the other hand, says Lustri, continuous process can achieve a capacity utilization rate of more than 80 percent. One study by the University of Cambridge illuminated other benefits for OSD manufacturers adopting CM for high capacity production:
• Plant footprint reduction by 70 percent;
• CapEx reduction by 25 percent;
• Operating cost reduction by 30 percent;
• Yield improvement by 10 percent;
• More consistent quality; more controllable, repeatable processes.
For sustainability fans, Cambridge’s researchers also noted that adopting CM can reduce carbon emissions by 50 percent.
However, most of what is in the public record does not seem to point to the industry moving en-masse in any hurry toward CM process for OSD manufacture. However the industry’s technology suppliers are increasingly confident that the shift is occurring and that its general adoption is inevitable.
GEA Pharma Systems Kris Schoeters noted that continuous processing techniques are not as uncommon in the pharmaceutical industry as everyone seems to think. Schoeter believes there are a number of reasons why the industry has been quite slow to adopt the technology:
• General concerns about start-up and shutdown and waste related to these phases.
• Perception on limited use, for example, that it is only suitable for large volumes.
• Regulatory concerns.
• Installed base of existing (older) equipment in many companies, causing them to question why they should change.
He asserted, though, that the abovementioned concerns are not an issue with certain continuous processing technologies and that switching from traditional processes in general does not lead to a big regulatory impact.
In a recent Pharmaceutical Engineering contribution, Hector T. Davila, director Process/Specialty Engineering, Fluor Engineering explained that firms that act as an integrator of owners’ requirements, production constraints, business and capital objectives, find that a stochastic view of the implementation of a continuous process is an effective approach. He said these companies, including Fluor, have a distinct opportunity to look both at the micro and macro of the Integration process. In his article he offered some general observations on the state of development of continuous oral solid dosage manufacture:
• Integration of PAT at critical unit operations is critical to maintain quality and avoid product loss.
• Supplying a continuous process with a large volume of excipients requires more careful logistics planning and control at the warehouse level.
• Use of bulk bags requires modifications to standard warehouse design and planning.
• The overall Continuous Process response to individual unit operations hysteresis is a concern since the available equipment may not deliver proper response and turndown ratios.
• To support quick changeover, it is necessary to determine the application of CIP or dedicated change parts and components.
• Disparity in OSD Continuous Processing can often be traced to Equipment throughput and turndown.
• Equipment processing duration during a production run varies from step to step. The longest duration limits the overall line capacity.
• Pay attention to Transient operations and response to changes.
Rutgers’ Oka and Muzzio also are sensing the inevitability of it all as well: “If recent investments by large Pharmaceutical companies in researching and developing continuous processes for manufacture of solid oral dosage forms are any indicators, then we are very likely to see rapid change in this arena, taking much less than 25 years.”
Interestingly, say Oka and Muzzio, some of the unit operations that are involved in the process of making tablets and capsules today are intrinsically continuous in nature, namely, milling, spray drying, roller compaction, extrusion, capsule filling tableting and packaging. “This implies that an existing batch process can be transformed into a continuous process with relatively minimal change in most processing steps.”