Advances in pH Measurement in High-Temperature Biotech Processes
pH is one of the most important parameters of cell-culture and fermentation processing in the biotech industry. Yet pH measurement in various industrial processes still heavily relies upon century-old pH glass electrode technology, due to its reliable performance and robustness.
However, with growing demand for increased accuracy of pH measurement and operational life of sensors, new technologies are emerging—especially regarding sensor durability in high-temperature steam-sterilization processes.
This article presents recent research on the aging mechanism of the pH-sensing glass after hightemperature exposure by using the non-destructive complex impedance spectrum method. The aging behavior of new high-temperature glass formulations in steam-sterilizable sensors, as well as the conductivity dependence of alumina-based reference junction potential, are discussed in comparison with standard steam-sterilizable pH sensor formulations. The article also introduces a more robust, built-in temperature sensor design for steam-sterilizable pH sensors.
Glass Aging in a High-Temperature Environment
All users know that pH glass electrodes age much faster in high-temperature environments. A typical symptom of an aged pH-sensing glass is its sluggish response toward pH changes. Understanding the aging mechanism is important in slowing the aging process of a pH-sensing glass. In this study, the complex impedance analysis method is used.
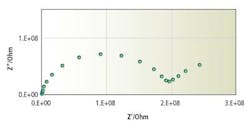
The method is non-destructive and has been widely used in studying electrochemical processes of an electrode. Figure 1 shows a typical complex impedance spectrum of a pH glass electrode with the real component of the impedance assigned to the abscissa and the imaginary component to the ordinate. At a high-frequency region, a semicircle is observed; and at a low frequency region, a declined line resembling a constant phase element is observed.
Figure 1. Impedance spectrum of a pH-sensing glass with the frequency range of 1 KHz to 0.01 Hz and 100 mV excitation
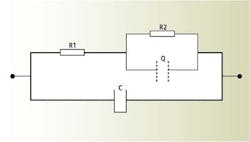
Analysis and simulation of the impedance spectrum yield an equivalent circuit of the pH-sensing glass as shown in Figure 2, where C is the capacitance formed by the pH glass membrane as the dielectric; R1 is the electric resistance of the bulk layer of the pH glass membrane; R2 is the electric resistance of the gel layer; and Q is the constant phase element of the gel layer representing the transportation properties of ions through the gel layer, which are frequency dependent.
Figure 2. Equivalent circuit of the pH-sensing glass
By comparing the equivalent circuit in Figure 2 and the impedance spectrum in Figure 1, it is clear that C and R1, which are reflected by the semicircle at the high-frequency region, represent the properties of the bulk body of the sensing glass; and R2 and Q, which are reflected by the declined line at the low-frequency region, represent the properties of the surface gel layer of the sensing glass. Since the complex impedance analysis is a nondestructive method, the same pH glass electrode was exposed to an aging process at 110º C for seven days.
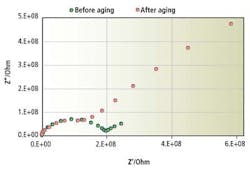
Figure 3. Complex impedance spectra of a glass electrode before and after an aging process at 110º C for seven days
Figure 3 compares the impedance spectra of the glass before and after the aging process. From Figure 3, it is clear that the main change of the glass after the aging process is with the declined line at the low frequency region, which means that the aging process has an impact mostly on the surface gel layer of the sensing glass. The value of all the components in the equivalent circuit can be obtained through simulation of the impedance spectra.
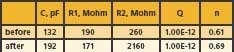
Table 1. Simulation value of the equivalent circuit of a glass electrode before and after the aging process
Table 1 summarizes the value of all the components in the equivalent circuit before and after the aging process. As shown in Table 1, R2, the surface gel layer resistance, had the most impact after the aging process. Further understanding of the gel layer resistance is the key to improving the durability of the pH-sensing glass against aging in a high-temperature condition.
Steam Sterilization Cycle Test
As shown above, the increase in gel layer resistance of a pH-sensing glass R1 is responsible for its aging. Since both Q and C are frequency dependent, a dc resistance measurement will reflect the change of R1, indicating the aging process of the measured pH-sensing glass. To compare the aging process, three groups (A, B and C) of pH glass electrodes were tested.
The A electrodes were made from a new high-temperature glass formulation based upon the research showing that aging occurs primarily in the gel layer of the sensor. The new formulations are designed to achieve exceptional thermal resistance at temperatures as high as 145º C. B and C sensors were supplied by manufacturers of existing steam sterilizable equipment.
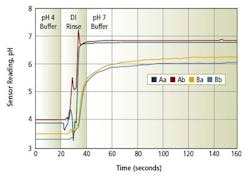
All the glass electrodes went through steam sterilization cycles of 30 minutes at 130º C. After each steam sterilization cycle, the dc resistance of the glass electrode was measured at ambient temperature. The A group of pH glass electrodes changed the least among all the electrodes tested, indicating that the new high-temperature formulation ages slower than the other pH glasses tested. One of the main symptoms of aged glass is sluggish response. To verify the finding, the pH response curve of the B-type pH sensor and the A high-temperature sensor were measured after a 50-cycle steam sterilization process.
Fig. 4. pH response curves of sensors after 50 steam sterilization cycles
Figure 4 shows the measured pH response curves of the sensors. After 50 cycles of steam sterilization, the B-type sensors showed typical slow response to pH changes, while A showed a response speed almost equivalent to non-aged pH sensors. The 95 percent response time of the A-type sensors after 50 cycles is about 10 seconds, while that of the B-type sensors is more than one minute.
Reference Junction Potential of New Steam-sterilizable pH Sensors
The reference system of a pH sensor is as important as the pH glass electrode where the stability of the sensor output is concerned. Almost all pH sensor manufacturers use Ag/ AgCl electrodes as the reference, because they are well known for their stability. However, the most uncertain component in a reference system is the reference junction, which separates the reference fill solution and the sample solution.
An ideal reference junction will allow both the cation and anion of the KCl reference fill solution to diffuse out evenly and generate zero potential at the junction. However, depending on the material of the junction and the ionic strength of the sample solution, there will be an uncertain junction potential built up, which can cause errors in the measurement. The new steamsterilizable pH sensor uses a SiO2/ Al2O3-based porous ceramic as the junction material.
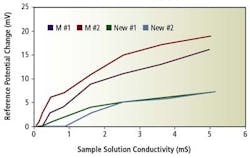
Junction potential studies indicate that this material exhibits the least electrochemical activity as a reference junction. Figure 5 shows the change in reference potential with the variation of the conductivity of the sample solution. The test started with deionized water as the sample solution. The conductivity was changed by adding a phosphatebased pH 7 buffer.
Figure 5. Dependence of reference potential on the sample solution conductivity
Since the conductivity of the sample solution is the only variable in this test, the change in reference potential is purely caused by the reference junction. From Figure 5, it is clear that the new sensor reference junction is more stable than the other group at the typical conductivity level of 5 mS/cm. The stability guarantees a more accurate pH measurement in a process when the ionic strength fluctuates.
Built-in Temperature Sensor Installation
Built-in temperature measurement of the steam-sterilizable pH sensor is an increasingly popular capability. Most designs of pH sensors place the tem-perature sensor in the sensing glass chamber of the sensor. Most pH sensor instruments use a non-isolated circuit design to handle both pH and temperature measurements. Such a design compromises the robustness of the pH sensor due to the addition of the temperature sensor. In other words, the deterioration of the temperature sensor isolated in the sensing-glass chamber during aging will directly affect the pH measurement.
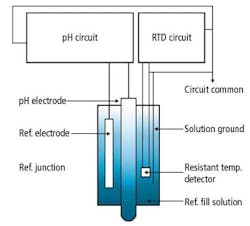
Figure 6. Block diagram of a new pH sensor with built-in temperature sensor
The new steamsterilizable pH sensors, however, use a design as shown in the block diagram of Figure 6. With the temperature sensor (RTD) installed in the reference chamber and a solution ground in the same chamber tied to the circuit common, the temperature sensor will not have any impact on the performance of the pH measurement.
Conclusion
Glass electrode pH sensors have been the workhorse of the biotech industry for decades, but the new demands of biotech processes are showing the weaknesses of this technology. New research is showing the way to advanced capabilities in sensor glass, reference junction technology and temperature compensation that respond effectively to the requirements of biotech processes with steam sterilization and other challenging environments.
About the Author
C.D. Feng serves as senior manager of sensor technology for Emerson Process Management, Rosemount Analytical Liquid. He has contributed more than 20 papers to industry publications on various topics. In addition, Dr. Feng has been awarded six U.S. patents, and received two ISA-GTF awards. He recently won the 2007 ISA Arnold O. Beckman Award for the research and development of advanced electrochemical sensing and instrumentation. Dr. Feng received his bachelor’s bachelor’s degree in chemistry from Shandong University in China, his master’s degree in material science from the Chinese Academy of Science, and his doctorate in material science from Nagasaki University in Japan.