As pharma companies expand, they are looking more and more to biologics for their next potential blockbusters. However, this class of product — ranging from well-established large molecule drugs to truly novel therapies — poses major challenges because of its scientific complexity and sophisticated development requirements.
Furthermore, expanding the drug pipeline isn’t the only growth strategy most companies are pursuing. They are also planning to expand geographically and expect to face various risks doing so, including unfamiliar regulatory environments, shifts in pricing and changes in customers’ ability to pay.
All this means that risk management is rising in pharma executives’ agendas. To manage risks, companies are developing strategies that involve both building internal capabilities and reliance on external expertise.
PRODUCT GROWTH
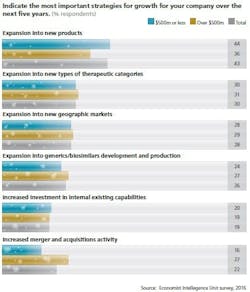
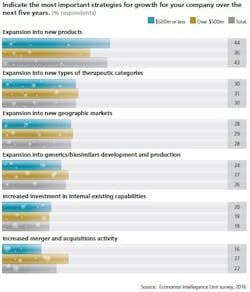
A March 2016 multinational survey from the Economist Intelligence Unit (sponsored by MilliporeSigma) titled, “The Changing Biopharma Risk Equation” of 254 pharma executives found that companies are pursuing different classes of new biopharmaceuticals as part of their expansion.
“Strides are being made in rare diseases and orphan drugs, as well as autoimmune disease,” says Andrew Baum, managing director of equity research, Citi. “And with Immuno-Oncology, you have a growing number of drugs with known efficacy in multiple indications.”
These developments are quickly translating into profits. Sales of biologic products — which employ sophisticated bioprocessing technologies in their manufacture and are used to treat a host of chronic diseases including cancers, diabetes and arthritis — are rising sharply and expected to grow from a $162B market in 2014 to $278B by 2020.1
Many of the new therapies help to address conditions that previously had no significantly effective treatments; the demand for such biopharmaceuticals has been so insistent that these new drug therapies have received significantly more U.S. FDA approvals in the 2015 calendar year than the average number approved annually over the last decade. It is not surprising, then, that biologics are a rising priority for most pharmaceutical companies surveyed.2
Indeed, the survey shows that stem-cell-derived therapies and gene therapies top the list of drug categories deemed likely to disrupt short- and long-term corporate strategies. However, nearly half (48 percent) of survey respondents indicated that they themselves are considering or are already in the process of developing novel therapies. These newer therapies are taking a greater share of production focus than more traditional drug products such as vaccines (38 percent) and blood-derived products (32 percent).
PRODUCT DEVELOPMENT RISKS
Risks of developing entirely new types of drugs are not new to the pharma industry. Since survey respondents highlight cell therapies as the category most likely to disrupt corporate strategy, it follows that the majority (94 percent) of respondents see the development of new and different drug products as increasing the importance of risk management. Those risks include new science and scarce funds for development and revenue.
After regulatory uncertainty (32 percent), a lack of funding for growth emerged as the second biggest concern (25 percent) for survey respondents overall. The inherent complexity of manufacturing diverse types of biologics requires relatively more funding than traditional therapies.
Complicating the funding equation for new drugs and novel therapies are the headline debates over drug pricing in general, which are at their most intense in the U.S., where double-digit drug price rises have been the focus of congressional hearings. “Pricing is the major concern,” says Baum in the EIU report, “because that increases systemic risk and creates a lot of bad will — and creating bad will in a heavily regulated industry is not a good thing.”
GEOGRAPHIC GROWTH
This focus is notable given emerging markets’ somewhat rocky overall economic performance. Asia came up as a particularly attractive region for the pharmaceutical sector in the next five years, with higher shares saying that they expect to be operating in many countries five years from now than say that they have current operations there: Indonesia (34 percent currently operating vs. 40 percent anticipate operating), South Korea (34 percent vs. 44 percent) and Taiwan (30 percent vs. 42 percent).
Indeed, the survey found high levels of optimism for emerging markets’ potential overall. For every emerging market that respondents say they anticipate entering in the next five years, at least half of respondents also say that they anticipate return on investment associated with entering emerging markets to increase in the next five years.
GEOGRAPHIC RISKS
Pharma companies have been operating to some degree in many countries for decades. The geography-related risks they see now to their favored growth strategies and those they expect to be the most important five years from now largely include regulatory and political concerns.
Among emerging countries, respondents most often indicate they are currently operating in Brazil, China and India — all are nations with somewhat risky regulatory environments that involve various levels of complicated mandates. In China, for example, Ralph Marcello, principal, Deloitte Consulting’s life sciences consulting practice, states in the EIU report that he sees a shift away from investment as a result of increased compliance risk, regulatory issues and price pressure on the Chinese national drug formulary.
It’s not just existing regulations that can be risky for companies expanding geographically, however — there’s also the risk of regulations being changed. A full third of respondents highlight regulatory uncertainty as potentially disruptive to their company’s strategy over the next five years. Adding the manufacture of new classes of untested biologic therapies in countries with unfamiliar or changeable regulations presents a high hurdle for companies considering that form of expansion.
But there is reason to be optimistic. Many emerging markets are standardizing their pharma regulations and, in some cases, aligning them with global standards.
MANAGING RISKS
To manage the risks which include regulatory, cultural and funding risks, companies most often say they will be addressing them by building internal capabilities and collaborating with outside experts.
For most, the risk-management strategy will involve building internal capabilities and business units (56 percent), with the second-biggest group pointing to use of outside experts such as contractors and consultants (51 percent). As always, there are major trade-offs to consider in the decisions companies make about whether to focus on in-house resources or to look externally, as well as tasking internal resources to manage and take input from external partners.
In addition to straightforward outsourcing, a range of partnerships is also important to companies; forming local partnerships emerges as the third most popular strategy cited in the survey (42 percent).
“When it comes to product innovation we’re seeing a greater willingness to use open innovation, collaborations and partnership with smaller companies, academic institutions or mid-sized companies,” says Marcello. “Companies recognize the majority of innovation no longer comes from inside the walls of a large biopharma.” And sometimes the more novel the therapy, the more important it is to include a broad range of insights in the innovation process.
Pharmaceutical executives are, on the whole, bullish about the next five years. Most report that they have a balanced portfolio of growth plans and strong confidence that they can overcome the expected risks.
As Baum noted, “in many ways, the industry has never had it so good.” However, to earn the returns they expect, pharmaceutical companies will need to build their internal capabilities and manage a range of outsourcing and partner relationships. They will need to learn to thrive in new cultures and ensure that their geographic growth is diversified enough to prosper even if individual countries present economic or regulatory hurdles. And, as always, at the core they will need new science to succeed.
For the full results and analysis of the Economist Intelligence Unit report, please visit www.gobeyondbiopharma.com.
REFERENCES
[1] Persistence Market Research, Global Market Study on Biopharmaceuticals: Asia to Witness Highest Growth by 2020
[2] U.S. Food and Drug Administration Center of Drug Evaluation and Research, Novel drugs 2015 summary