Meeting Global Pharmacopeia Requirements for Water Compliance
While the public considers municipal water to be “pure”, the pharmaceutical market considers municipal water (feedwater) just the starting point in producing pure water. Water is the most widely used excipient in pharmaceutical manufacturing, and pharmaceutical water is a multi-functional resource, crossing all disciplines in the pharmaceutical industry. Water is used as a raw material, solvent, ingredient, reagent, and cleaning agent, and is produced in a variety of “pure” forms.
Purified Water (PW), Highly Purified Water (HPW), and Water for Injection (WFI) used in pharmaceutical processes are produced on site from the local potablewater, which has been produced by the treatment of the feedwater.
Today’s pharmaceutical companies have invested considerable capital in state-of-the-art instrumentation, purification equipment, storage and distribution loops,and importantly in the calibration and certification of their water systems. By understanding water, its sources and impurities, and the capabilities and limitations of purification methods, a water system can be designed to meet not only pharmaceutical companies’ requirements but to meet global pharmacopeia regulations.
Source water requirements
“It is prepared from water complying with the U.S. Environmental Protection Agency National Primary Drinking Water regulations or with the drinking water regulations of the European Union, Japan, or with the World Health Organization’s Guidelines forDrinking Water Quality.” USP 35
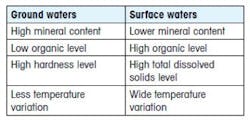
The feedwater source for a municipality can be from a surface water or a ground water supply. The impurities vary in each source and some of the primary differences are shown below:
Because the quality and characteristics of the feedwater supply have an important bearing on the purification, the pharmacopeias define the source water for the production of PW, HPW, and WFI. The pharmaceutical facility should communicate regularly with their water provider and request an annual water test report for the feedwater. To further the understanding of the feedwater and what technologies are required to purify it, below are the categories of contaminants found in a water supply.
Contaminants in feedwater
The impurities found in water can be categorized into six major classes: dissolved ionized solids, dissolved ionized gases, dissolved non-ionized solids (organics), particulate matter, bacteria/algae, and pyrogens. Feedwater varies significantly in purity both from one geographical region to another, and from season to season.
Total dissolved solids (TDS)
A measure of the total of organic and inorganic salts dissolved in water, obtained by drying residue at 180°C. The sum of all ions in a solution is often
approximated by means of electrical conductivity or resistivity measurements. TDS measurements are commonly used to assess reverse osmosis unit
performance.
Total ionized solids and gases
Concentration of dissolved ions in solution, expressed in concentration units of NaCI (sodium chloride). This determines the operating life of ion exchange resins used in water purification, and is calculated from measurements of specific resistance. Gases (carbon dioxide and oxygen) affect the water quality and system performance.
Total solids
Total solids in water include both dissolved and suspended solids. The quantity of total solids is determined by weighing a sample of the feedwater before and after evaporation.
Microbial
Bacteria, viruses, and pyrogens (endotoxins).
Particulates
Sand, dirt, and decay material.
Organics
Organic matter is a broad category that includes both natural and man-made molecules containing carbon and hydrogen. All living matter in water is made up of organic molecules. The most common are by-products of vegetative decay such as tannins, lignins, and humic acid. By knowing the variety of contaminants in the water and the removal capabilities of the different available purification processes, a system can be designed that will produce the water quality required for a pharmaceutical facility. There are a range of purification technologies and we have provided below a brief description of the major purification technologies.
Major water purification technologies
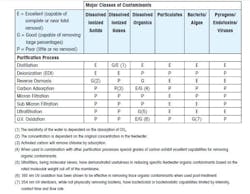
The chart shown below is a summary of the removal capabilities of different purification technologies versus the contaminants commonly found in water.
PW, HPW, and WFI for pharmaceutical use are produced via a combination of different purification technologies. As with the source water, each pharmacopeia defines the methods of production, but for PW and HPW the technologies utilized is the decision of the system designer, with the only requirement being that the water meets the pharmacopeia regulations for quality. For WFI, to meet the pharmacopeia requirements (all pharmacopeias except Japan), the final purification process must be distillation.
Purifying the feedwater for use in the pharmaceutical industry requires a series of steps. The objective is to remove the impurities in the feedwater while minimizing additional contamination from the components of the purification system, the storage tanks, the distribution system, and from possible biofilm growth. Selection of the correct purification technologies and the instrumentation to monitor the system are critical to success.
Reverse osmosis
Reverse osmosis is best understood when related to osmosis itself. In one of the experiments performed by everyone in first year chemistry, a semi-permeable membrane (a membrane that is permeable to water but not to salt) is used to separate two solutions; a saline solution and pure water. The pure water will flow through the membrane to dilute the saline solution. This is osmosis. When pressure is applied to the sa-line solution, the natural process of osmosis can be overcome and even reversed. With sufficient pressure, pure water can be forced out of the saline solution through the membrane and into the pure water side of the vessel. This is reverse osmosis.
In reverse osmosis for pharmaceutical water production, a membrane is also used for the separation of contaminated water. Membranes can be made from cellulose acetate, polyamide, polysulfone, or a variety of proprietary formulations. Two configurations are common: “hollow fiber” and “spiral wound”. Hollow fiber membranes look like a group of drinking straws gathered into a bunch, the spiral wound resemble a helix.
Because the quality of water produced by a reverse osmosis apparatus is directly dependent upon the quality of the input water and because effective removal of ions rarely exceeds 97%, reverse osmosis is widely used as a pretreatment process to purify feedwater before introduction into an ion exchange unit or a distillation system.
Distillation
Distillation is the oldest form of water purification and has been utilized by humans since we first boiled wa-ter in a cave. It is a unique process because it removes the water via a phase change and leaves behind the impurities. In distillation, water is heated to itsboiling point and undergoes the first of two phase changes, from a liquid to a vapor. The solid ionic ma-terials, the particulates, the microbials, endotoxins, and most of the dissolved organic contaminants are left behind in the boiler. The pure steam is then passed through a cooling coil where it undergoes a second phase change from a vapor back to a liquid. For the production of WFI, the pharmaceutical distillation system is normally fed water that has been pretreated by a variety of other technologies. The pretreatment is used to reduce the costs of maintenance on the distillation system and to ensure the quality of the distillate. Distillation is the only purification method that removes 100 percent of biological materials whether bacterial, viral, or pyrogenic.
Deionization
Deionization or ion exchange is a process also mistakenly called demineralization. The Encyclopedia of Chemical Technology defines deionization as:
“The reversible interchange of ions between a solid and a liquid phase in which there is no permanent change in the structure of the solid.”
Deionizers are generally available in two forms: a two-bed and a mixed-bed configuration. In the two-bed configuration, the cation and anion resins are in two discrete columns or in two discrete layers in the same column. The advantage of the two-bed deionizer is that it can purify a greater volume of water than a comparable mixed-bed system; however, they produce lower quality water.
The mixed-bed deionizer contains an integral mixture of anion and cation resins packed in a single column. Only mixed-bed deionization can produce water with a resistivity of 18.178 million ohms, which is theoretically ionically pure.
Ion exchange technology is designed to remove ionized or charged material from water. Even though water will be ionically pure after the deionization process, the water will still contain non-ionized solid and gaseous materials (organics), bacteria, viruses, and pyrogens. These are not ionically charged species and cannot be removed by ion exchange processes.
Electrodeionization
Electrodeionization (EDI, also known as EDR, CDI, and CEDI) is a technology that combines ion exchange resins, ion-selective membranes and an electrical current to remove ionized contaminants from the water. Reverse osmosis is typically used before EDI to ensure that the EDI stack is not overloaded with high levels of salts. Usually, reverse osmosis removes about 97% of ions. EDI will remove 99% of the remaining ions as well as carbon dioxide, organics, and silica. In electrodeionization, the water passes through multiplechambers filled with ion exchange resins held between cation or anion selective membranes. Under the influence of an electrical field, the anions and cations migrate across the membranes to the anode and cathode. Typically, EDI product water has a resistivity of 11 to 18.2 MΩ-cm (at 25°C) and a total organic carbon (TOC) content below 20 ppb. Bacterial levels are minimized because the electrical conditions within the system inhibit the growth of microorganisms.
Carbon adsorption
In adsorption, the organic impurities in water form a low-energy chemical bond with the surface of activated carbon. Because adsorption is a technique for removing only organics and chlorine, it is most often used as a pretreatment to remove large amounts of organic impurities prior to other purification processes. Activated carbon is very effective at removing chlorine and other oxidants at rates of 2 to 4 times the chemical weight of the oxidant. By removing the oxidants, the opportunity for microbial growth is increased and must be controlled and monitored.
Ultraviolet light
Ultraviolet light at the 254nm wavelength is used asbactericide. This wavelength disrupts the ability of bacteria to reproduce. UV at 185nm will break down organic contaminants to CO2 and water for subsequent removal by ion exchange.
Filtration
Filtration can be performed by one of two methodologies, either depth filtration or membrane filtration. Depth filters can be made of sand in a container or of fiber wound around a core. Both methods mechanically strain out sediment and particulate matter.
Membrane filtration, on the other hand, is physical straining by a single layer of membrane material. The membrane material is produced from man-made resins and can be either hydrophobic or hydrophillic. The pore size is tightly controlled and therefore absolute removal of particulates with diameters larger than the pore size can be achieved. In pharmaceutical systems, filtration is normally limited to the pretreatment section because although filters trap contaminants, it is possible for bacteria to pass through a membrane filter.
Conclusion
Once the feedwater source is known and the purification technologies have been selected, knowing what parameters need to be utilized to control and monitor the system are critical. By understanding feedwater and the water purification system, a consistent supply of Purified Water, Highly Purified Water or Water for Injection can be ensured.
Download the full Pharmaceutical Waters guide from Mettler Toledo here