Design of Experiments Helps Increase Yield of Pharmaceutical Intermediate from 70% to 88%
Researchers at Codexis Laboratories Singapore performed a full-factorial designed experiment with 20 runs to determine the impact of four independent variables on product selectivity during a silylation reaction. The experiment revealed that the optimal combination of factors increased the selectivity of the reaction to 97% for silylation on oxygen with less than 2% each for the undesirable alternatives. Codexis researchers adjusted the factors to increase the concentration for improved throughput, resulting in a process that delivered 95% selectivity along with an 88% yield.
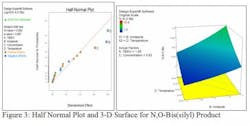
A joint research program between Codexis and a leading pharmaceutical manufacturer addressed the production of an O-silylazetidinone, a key intermediate for the synthesis of many beta-lactam antibiotics including ertapenem, meropenem and doripenem, through the silyation of a hydroxyazetidinone. The output of the original process consisted of 70% the desired product where the silyl group is on oxygen. The output also included 11% of an isometric byproduct where the silyl group is on nitrogen and 17% of a third option where the silyl group is on both oxygen and nitrogen (i.e. bis-silylation). Codexis develops and commercializes processes to active pharmaceutical ingredients (APIs) and pharmaceutical intermediates using its proprietary biocatalytic processes. The overwhelming majority of APIs have at least one chiral center. Increased regulatory requirements for improved product purity have led to growing demand for stereochemically pure intermediates. Using proprietary biocatalysts, Codexis manufactures virtually 100% chirally pure intermediates and APIs. These are often manufactured directly at high purity, compared to low purity materials that must be isolated after being produced by traditional chemical processes.Codexis has developed an efficient and selective process for dynamic ketone reduction of a ketotester to produce a diastereomeric hydroxyketone. Conversion of the diastereomeric alcohol into the O-silylazetidinone requires additional challenging steps including the silyation of the hydroxyazetidinone. The previous pilot plant process involved treatment of a slurry of hydroxyazetidione in toluene at 25-30oC with imidazole and then feeding in a solution of tert-butyldimethylsilyl chloride (TBSCI) in toluene. After workup and crystallization, this provided the desired product in yields of about 70%. A team of Codexis researchers including Steve Collier and Rob Wilson, set about scaling down the current process in the laboratory, addressing the selectivity and yield issues and transferring the process back to the pilot plant in as short a time as possible. The conventional approach to optimizing the factors would have been to run a series of experiments while varying a single factor, sometimes called the one-factor-at-a-time (OFAT) approach. The problem with this approach is that it does not detect interactions between factors or second order effects.Researchers decided to use the design of experiments (DOE) method because it varies the values of all variables in parallel so it uncovers not just the main effects of each variable but also the interactions between the variables. This approach makes it possible to identify the optimal values for all variables in combination. It also requires far fewer experimental runs than the OFAT approach. “Design of experiments enables chemists to efficiently define, better understand and optimize factors that are important to reaction yield and robustness, particularly where multiple parameter interactions occur,” Collier, who is Director of R&D for Codexis Laboratories Singapore, said.The Codexis team uses Design-Expert® software from Stat-Ease, Inc., Minneapolis, Minnesota, to design and analyze DOE experiments. “Design-Expert walks users through the entire process so it enables scientists and engineers to develop their own experiments without requiring assistance from statisticians,” Collier said. “I am an advocate of DOE and a fan of Stat-Ease so I brought one of their instructors in to give my team a course in DOE. It was very well received and we have since utilized Design-Expert to help improve a series of important pharmaceutical processes.”The Codexis team selected four factors in the O-silylazetidinone process for their potential to have a significant impact on the process: equivalents of TBSCI, equivalents of base, reaction concentration and reaction temperature. The team developed a two-level factorial DOE study with 20 runs including four runs at the existing conditions in order to evaluate the robustness of the experimental data. Three responses were studied including the yield of the desired O-silyl compound, yield of the undesired N-silyl compound and yield of the undesired N,O-bis silyl compound after completion of the reaction.The experiments were centered around the existing conditions which were 1.1 equiv TBSCI, 1.2 equiv imidazole, 0.868 N concentration and 30oC temperature. The additional runs were designed using small changes in either direction from the existing conditions. The results of each run were quantitatively analyzed using high performance liquid chromatography (HPLC). The HPLC data was analyzed using Design-Expert and models were built to determine the impact.
The DOE models showed that high levels of imidazole and high temperatures tend to increase the yield of the desired O-silyl product and that these conditions also reduce the yield of the undesired N-silyl byproduct. “The power of DOE comes from its ability to identify interactions between factors,” Collier said. “In this case, the temperature and imidazole loading had a simple additive effect but in many other examples the interactions between factors are much more complex. Understanding these interactions, which cannot be detected without DOE, is critical to optimizing the manufacturing process.”The best selectivity was provided by a run that yielded 96.6% O-silyl, 1.9% N-silyl and 1.5% N,O-bis(silyl). However, this run had a relatively low concentration so its volumetric productivity was not at optimal levels. Based on the information provided by the DOE, further experiments were performed by using the factor values that were demonstrated to provide high selectivity for O-silyl while increasing concentration in order to improve throughput.“We knew if we ran dilute we could optimize selectivity but at the same time we would be reducing volumetric productivity,” Collier said. “Fortunately, the DOE had already shown us what levers to pull to increase selectivity. As we increased the concentration, which is negative to selectivity, we also increased the base and temperature even further than in our DOE runs to compensate. In the end we achieved the same selectivity we had observed in the DOE runs at a higher level of concentration, providing the yield that we needed for high volume manufacturing.”The final conditions utilized 1.05 equiv TBSCI fed to the reactor over 1 hour, 1.3 equiv imidazole, a reaction temperature of 50 C and a concentration of 0.868 M. At the lab scale these conditions generated 96.9% O-silyl product with only 0.8% N-silyl and 0.8% N,O-bis(silyl) byproducts and 1.5% unreacted starting material. Execution of these conditions on a 10 g scale provided the desired product in 85% isolated yield with a further 8% material remaining in the mother liquors.These conditions were then successfully transferred to Codexis’ partner for further development and implementation at pilot scale. Scale-up proceeded smoothly and the process was run in the pilot plant using 58.2 kg of substrate providing a yield of 88.5% with an assay purity of 95.4%. The entire process described in this article from receipt of the process to tech transfer back to the pilot plant was accomplished in only four weeks. “The dramatic improvements in the selectivity and yield that were achieved in this application combined with the short turnaround time demonstrates the power of carefully designed DOE studies,” Collier concluded.For more information, contact: --Codexis, Inc., 200 Penobscot Drive, Redwood City, CA 94063. Ph: 650.421.8100, Email: [email protected] , Web site: http://codexis.com-- Stat-Ease, Inc., 2021 E. Hennepin Avenue, Ste. 480, Minneapolis, MN 55413-2726. Ph: 612-378-9449, Fax: 612-746-2069, E-mail: [email protected], Web site: http://www.statease.com