Light Induced Fluorescence as a New Tool for Pharmaceutical PAT
By Paul Davies, Expo Technologies, St. Louis, Mo.
Light Induced Fluorescence (LIF) has been talked about lately as a potential new tool for the PAT toolbox. (See “QbD Tools: LIF Makes a Comeback.”) The recent IFPAC 2011 conference in Baltimore included a number of presentations showcasing this “new” technology. But can the technique really be labeled as new?
The first publication referencing work using LIF, with respect to pharmaceutical material, dates back to 2001. Work done by Lai, et al at the department of Chemical Engineering, Massachusetts Institute of Technology, in conjunction with the Consortium of Advancement in Manufacturing of Pharmaceuticals (CAMP), first demonstrated the technique’s application with pharmaceutical material.
But the LIF sensor used by the team at MIT is almost unrecognizable, compared with commercial units available today. Firstly, this early sensor utilized a flash lamp source. Flash lamps frequently show pulse-to-pulse intensity fluctuations. They require significant power to operate and may have limited capabilities to dynamically adjust the pulse rate or duty cycle. In addition, although the flash lamp source meant the unit had a broad excitation range through the UV and visible regions of the spectrum, a series of manually interchangeable narrow band pass filters were required in order to select the excitation wavelength.
In this early design, the detector was a solid state photomultiplier tube (PMT). The broadband sensitivity of the PMT necessitated a design which included a narrow band pass filter for the desired fluorescent emission wavelength.
These limitations may have played a role in the barrier to general acceptance and adoption as a routine PAT tool. But despite the practical implementation limitations, the analytical performance of these early units showed promise. Standard samples containing 0.1% active ingredient Triamterene, made into to standard tablet wafers, demonstrated a measurable signal.
With an estimated 50-70% of small molecule active pharmaceutical ingredients exhibiting fluorescence, it was no surprise that some of these early units were adopted by PAT thought leaders. But with such early promise demonstrated, why did the technique not become a staple in the PAT armory? A number of factors may be attributed. The form factor of these early units may have played a part, but the biggest challenge may well have been lack of choice of commercial systems. There were certainly many fewer choices when compared with another three letter acronym technique, namely NIR.
There are myriad NIR spectrometer system developers, vendors and technologies available from the NIR portfolio. From FT-NIR to Diode Array and even Micro-Electro-Mechanical Systems (MEMS), they all have their place. In the last decade, the development of NIR has outstripped that of LIF. With NIR having wider applicability, it is not difficult to understand why.
Rebirth
But as drugs become more potent, it is clear that NIR is struggling to detect the API levels desired. Hence the “re-birth” of LIF and with some new tricks in its bag, due, in part, to availability of new technology. Firstly, no longer is the continuum source flash lamp used, but instead, narrow bandwidth LEDs are employed. The development of high power, short wavelength LEDs covering the 240 nm to 400 nm ultraviolet/visible spectrum makes them an attractive alternative source. The LEDs feature low power consumption, desirable for battery powered applications such as blend monitoring, easily modulated pulse widths and duty cycles and easily controlled stable output powers. In addition there are now options to provide a fluorescence spectrum rather than just a fixed wavelength uni-variate measurement. The latter improvement is significant for a couple of key reasons. Firstly, a full spectrum measurement permits peak shifts as a function of concentration to be observed and not mistaken as pure concentration changes. In addition, a full spectrum measurement may permit more than one analyte to be measured simultaneously. Secondly, and more importantly, this allows the implementation, the form factor and data treatment, to mimic that of its big and widely implemented brother, NIR. Analyte concentrations as low as 0.01% may be observed with these new LIF technologies.
So what are the drawbacks of LIF? The main issue is that the analyte must exhibit fluorescent properties. In addition initial feasibility tests must be conducted to determine the optimal excitation wavelength for the analyte(s) to be measured. The instrument is then finessed to optimize the range of concentrations to be measured and to avoid any possible interference from other materials.
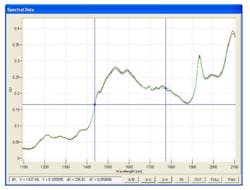
But the data from these next generation LIF systems look promising. Figure 1 shows typical NIR spectra of powder blends of caffeine in lactose, at varying concentrations between 0.05 wt% and 3.0 wt %.
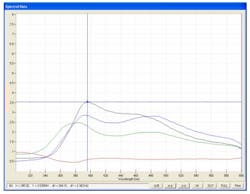
The fluorescence spectra, of the same powder blends, are shown in Figure 2. The sensitivity advantages are clearly shown.
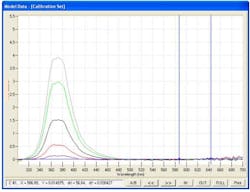
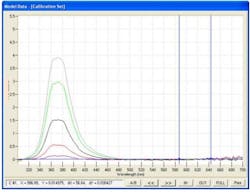
An additional benefit is the lack of fluorescent activity in most of the commonly used excipients (Fig 3.) such as magnesium stearate, microcrystalline cellulose and fumed silica.
In conclusion, where the LIF technique may never replace NIR, it should certainly be considered as a complementary tool in the PAT toolbox.