Building a Modeling Culture in Manufacturing at Pfizer
As the world's largest research-based pharmaceutical company, Pfizer has had great success using modeling and simulation to predict and improve the results of strategic initiatives within research and development. The research organization has been using simulation, specifically Discrete Event Simulation (DES), for more than 20 years, and it has become ingrained in the R&D decision-making culture.
The adoption of DES within the manufacturing organization has been less centralized and less systematically applied to the decision-making process. There have been many successes applying simulation to manufacturing projects, but they have been isolated. Sharing these DES projects and what is learned with the whole technical organization is an important next step. [For Cancelarich's ideas on what modelers shouldn't do, see "Modelers Gone Wild: Six Practices to Prevent."]
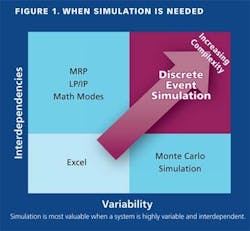
Our Operational Excellence Group has done a great job of communicating, particularly through Manufacturing Lean and Six Sigma initiatives, how variation can impact all areas of the organization, Pfizer Global Engineering (PGE) has furthered this understanding through the use of simulation and modeling, so that Manufacturing understands how variation and decisions based on averages can often lead to uncertainty and surprises in operations (Figure 1).
Challenges to Adopting Simulation Modeling
As recently as a few years ago, we did not have the readily accessible hardware—i.e., desktops and laptops—and computing power now available on most engineers’ desks. The availability and variety of software tools has significantly improved over the last 15 years and model development requires less specialized programming experience.
The right mix of tools is important, but the challenge of developing a simulation/modeling culture is the biggest barrier to the application of simulation in many organizations. Many manufacturers may go about building this culture in the wrong ways.
Often, a good first step is understanding that, in Manufacturing, the combination of time and the variability of inputs leads to levels of complexity that cannot be easily explained in a spreadsheet. We are very comfortable developing complex spreadsheets today, but they have their blind spots. If they are not understood, a team will move forward based on insufficient data and understanding, which can lead to manufacturing inefficiencies or other issues. Spreadsheets are immensely valuable—however, it is important to understand when simulation will bring the much deeper insight that will be necessary to succeed in today’s competitive markets. If a team does not know that such tools or resources are available, and how to use them, that opportunity will be lost.
Also it is important to understand that the simulation effort is a tool to help improve decision-making and not the final deliverable. In many cases it is assumed that simulating the facility or operation will solve all of our issues. But simulation is like all the other process tools we apply—another deep-reaching application that can deliver unique and valuable “objective” insights, so we can continue to curb and balance our subjective viewpoints.
The Value of Simulation for Pharmaceutical Manufacturing
Simply going through the process of developing a model can deliver critical insights into an issue or problem. Applying simulation often makes the team better, as it helps bring new and improved ways of looking at the process or manufacturing problem. Potential constraints can be challenged and risks can become clearer. Modeling itself does not solve problems; the creativity of the team is required for this. However, a new perspective can significantly improve the likelihood of an innovative solution or confirm that a true and tested solution is correct. Simulation also helps instill an understanding that issues and systems are not static and will change and continue to change over time.
It is important to accept that systems are changing and will not remain static. It is also critical to understand where a system’s tipping point is, and modeling and simulation help with this. Tipping points can include: not meeting supply commitments; resource constraints; not meeting design specifications; running a variance on budgets; poor inventory management; an expired product; or regulatory inspection actions. Staying as far away from a tipping point while managing costs and resources is the real challenge we deal with every day.
During model development, sensitivity studies are done in order to better understand the process and what variables are important. Watching how variables interact as one particular input is changed provides insights that cannot be gained by a spreadsheet. This activity can also help the team focus on a critical number of issues and not the complexity of the process.
The model does not need to look exactly like reality. It simply needs to best represent interactions between people, equipment, materials, and time. Often looking outside normal ranges helps the team understand if a variable is critical or not. The model can help instill a focus on a data-driven approach and not an anecdotal one. The greatest ability of simulation is to use data—either actual or estimated—to see where the interactions diverge and become critical.
Team should not be afraid to model with estimated data—it can really start the discussion and help the team understand the interactions. The data is easy to change. The value is in understanding the interaction and complexity generated by the interface of people, equipment, materials, and the rules and systems we put around them to control outcomes. Sometimes looking for all kinds of data or starting a heavy data collection exercise can become an easy deterrent. It’s possible to start simulation with data that is at hand today. When in doubt, teams should complete a sensitivity study around an unknown data set, helping to decide if more data is really necessary.
A Focus on Simplicity
Often when I use simulation to solve problems, these new insights bring more value than the initial computer model. Having teams develop simple models can often lead to new understandings on future issues and processes. Few leaders in an organization have the time and focus to understand all the interactions that a model can take into account. However, the solution had better be simple and easy to explain or the effort will be lost. In the end, that should always be the focus.
Communicating the importance of a given simulation solution should not be underestimated. The model can be used to facilitate this communication, but—especially when a clean, polished model is required—the focus can become the model and not the solution. If the focus becomes the model and why it does not “mimic” reality, communication can become difficult. It is often better to focus on how the critical issues interact, how the team came to its conclusion that they are critical, and how the solution resolves these interactions. The value is distilling the critical variables so options and solutions can be tried out and understanding how each solution can keep us from “tipping points.”
A team confident in its modeling solution is a team that will focus on the difficult task of communicating and selling the solution. It may take many 30-second elevator speeches. Development of direct, concise communication cannot be underestimated. In the end we need to get it right, but to make it happen we also need to be able to explain it right.
I am especially skeptical if someone says that they arrived at a solution because that is what the model said. As a practitioner of simulation, I know I can make a model do what I want it to. The real system does what the laws of nature and man say. However, the modeling effort when done correctly will give greater insights into those laws of man and nature—particularly how people, material, and equipment interact over time.
Tool Selection
Part of the challenge with using any type of technology is determining what tool is right for the job. There are many modeling tool sets currently available within Pfizer Global Engineering. This list represents what is currently used.
Process Fit and Analysis (static)
- Aspen Process Developer (BatchPlus): A recipe-based modeling technology for batch processes
- Intelligen SuperPro Designer: Facilitates modeling, evaluation and optimization of integrated processes. It provides modeling of manufacturing and end-of-pipe treatment processes, project economic evaluation, and environmental impact assessment.
- Intelligen SchedulePro / Production Modelling Ltd Orchestrate: Finite Capacity Modeling. They handle production scheduling, capacity analysis, modeling, and debottlenecking of multi-product facilities.
Thermodynamic and Rigorous Modeling
- Chemstations ChemCad: Suite of chemical process simulation software
- Aspen Suite and Aspen Plus (Aspen Chromatography, Dynamics, Reaction and Solubility Modeler): Facilitates conceptual design of chemical processes
Spreadsheet-based Business Modeling
- Oracle Crystal Ball: Suite for predictive modeling, forecasting, simulation, and optimization.
- Microsoft Excel-based tools
Discrete Event Modeling
- ProModel Optimization Suite: Discrete-event simulation tool that also allows modeling of continuous processes
- ProModel Project Simulator: Adds simulation capabilities and analysis to Microsoft Project files
- ProModel Portfolio Simulator: Allows modeling of portfolios
- ProModel Process Simulator: Adds simulation capabilities and analysis to Microsoft Visio flowcharts
All these tools have value and each has its own sweet spots. Understanding when to apply simulation and which tools to apply is an important first step. Very often it is critical to use multiple tools to flesh out the issues with a project or problem. One tool cannot solve everything—one becomes a technician with a hammer and all the problems start to look like nails. I recommend that any engineer understand when and how to apply simulation and be skilled in the different general sets above. We are working to enable this capability within Pfizer.
Many of the tools above do not require continual specialized training, but they can require some practice. There are common pitfalls most engineers go through at some time or another. Understanding when to apply simulation and where the modeling focus needs to be are special skills which are best acquired by a solid understanding of the tools and knowledge of manufacturing. In most organizations today, these skills are lacking. Establishing a culture of modeling, I believe, leads to deeper and more valuable applications of simulation, and fewer barriers to success.
The Need for Simulation Analyses in Manufacturing
Manufacturing systems by nature are highly variable. Every system has different levels of interdependency and variability. The more parts to a system, the more a system is influenced by each of those parts and the more interdependent it becomes. As the interdependencies increase, the variability also increases and the more unpredictable the systems become. This makes it increasingly difficult to understand and describe the behavior of the systems, let alone determine the outcomes. Using discrete event simulation in these situations provides a more realistic and objective picture and the ability to conduct more insightful analyses (and avoid the scenario in the image below).
Moving to a Manufacturing Simulation Culture
As one of the Engineering Directors within PGE, I want to see the benefits of simulation throughout our pharmaceutical manufacturing organization. We can benefit from a deeper knowledge of how our process and business systems are influenced by the decisions an organization regularly makes. How an organization plans and manages assets, resources, and materials, and the types of business processes in place, can be very complex and dynamic, particularly as conditions and inputs change with time. We need to get beyond the averaging of information and data, whether it represents capacity, equipment, or resource availability, and try to understand the more dynamic interaction over time. We need to better anticipate future manufacturing surprises and bring opportunities to light.
Our organization has benefited greatly from a focus on operational excellence and the enabling of simulation within PGE. I believe that we can continue to significantly improve by growing a manufacturing culture that is as comfortable with simulation as with spreadsheets. It will take time and development of professionals skilled in our modeling tools as well as our business processes.