Beware Thermal Cycling in BioProcessing Part One
Valves and fittings account for the largest number of seals used in any bioprocessing system. In an individual processing suite, the seals that make up sanitary connections to tanks and vessels, valve ends and upper works, and tubular fittings may number in the hundreds.
Throughout an entire facility, the number may be in the thousands. Reliability of the seals is a function of design, quality, material selection, and installation—all considerations made before the valve or fitting is subjected to the actual service.
However, once the components are in service, they are vulnerable to thermal cycling, a major factor that affects the reliability of the seals. This article examines the effect of thermal cycling on stem seal and seats in the ball valves that are used extensively in utility systems, as well as its impacts on the seal in sanitary clamp-type fittings used throughout both the process and the utility systems.
Its goal is to highlight valve and fitting design improvements in seal containment as well as ways to improve the control of loads applied to the seals both during initial make-up and thermal cycling. Fermentation is the basis of a bioprocess and includes both microbial and mammalian cell culturing.
Although similar, each has its own unique requirements. Microbial fermentation usually refers to large-scale cultivation of living microorganisms or single-cell creatures. Mammalian cell culturing involves growing complex cells that come from the organs and tissues of animals, which are much more fragile and more difficult to grow.
A complete system for manufacturing an active ingredient for a biopharmaceutical product is made up of tanks and vessels, pumps, centrifuges, and other rotating equipment, as well as various kinds of operation-specific equipment. Fluids are transferred from device to device by means of tubing, pipe, and hose.
Fittings connect all parts of the system together, and valves control the fluid within the system. The process system is supported by certain clean utility services, such as pure water, sterile air, and steam for sterilization. The seals in all of the equipment and subsystems and the clean utilities are critical to the reliable operation of the complete system.
The system consists of upstream preparation, fermentation, harvest and recovery, and purification and refining. Upstream preparation includes the preparation of media, the substrate and nutrient mixture that will be the environment in which the organisms will live and grow; buffering solutions, used to control the allimportant pH; and inoculum generation, preparation of the cultures that will be placed in the fermenter to begin the process.
The fermenter/bioreactor provides a contained and protected, controlled and homogeneous environment in which the microorganisms and mammalian cells reproduce and grow. When the fermentation process is complete, the broth is harvested and sent through a recovery process. If the product is extracellular, the cells are removed from the broth. If intracellular, the cells are disrupted and the debris removed.
Purification is the final downstream processing after recovery. During purification, the desired product is separated from the broth—using a combination of methods, including precipitation, filtration, and chromatography—refined, and concentrated. After use, each piece of equipment and each run of conduit through which fluid had been transferred must be drained, cleaned, and sterilized in preparation for the next production run.
Valves and fittings can have a direct effect on how effective the cleaning and sterilization process will be.
These components must be:
- completely drainable, leaving no entrapment areas or puddles where contaminants can accumulate;
- cleanable using current clean-in-place (CIP) methods;
- sterilizable, allowing all internal surfaces to be in contact with steam; and
- able to withstand the thermal cycling of repeated sterilization processes.
Seals must be leak-tight throughout the entire process. Leaks cannot be tolerated, since they could result, either in the release of potentially hazardous materials out of the system, or destroy sterile condition inside the system. Internal leaks can compromise the process, cleanliness, the sterile environment, and instrumentation and control procedures.
Clean Utilities
All bioprocessing system operations must be supported by clean utility services, including pure water, clean steam, and sterile air. These services are supplied to the various pieces of equipment via extensive piping systems, which are connected by welding or sanitary fittings.
Diaphragm valves are used; however, the diaphragm may have limited life in steam service, especially where the valves are actuated frequently. Ball valves are an accepted industry standard for isolation purposes on continuous pure/clean steam service (The American Society of Mechanical Engineers standard, ASME BPE Bioprocessing Equipment, is an American National Standard).
Generally, the service parameters—pressure, temperature, and flow—are not severe in these systems. The most difficult condition valves and fittings must withstand is thermal cycling. Thermal cycling impacts the durability of the plastic and elastomer seal materials and is a common cause of leaks.
Ball Valves
Ball valves have seals at the stem, seats, body, and at the connection into the system. The body seals are static seals, which are made up when the valve is assembled, and do not have to be cycled mechanically during service. Adequately contained, body seals normally can withstand thermal cycling without leakage, as long as the seal material is not degraded during the process.
If the manufacturers’ instructions are followed during maintenance, body seals should not pose a leakage problem. Construction personnel or technicians install the ball valves into systems when they are built or maintained. The end connections on the valves use the same kinds of fittings used to assemble the complete system and connect all the parts together.
Those connections will be discussed in Part 2 of this article (to appear in September’s issue of Pharmaceutical Manufacturing) on sanitary fittings. The critical seals in ball valves are at the stem and seat and are part of the basic design of the valve. They are dynamic seals and must retain leak-tight performance during and after both mechanical and thermal cycling.
Stem Seals
Stem seals are intended to prevent leaks into or out of containment. Leaks are driven by pressure and proceed from a high-pressure to a low-pressure region. In pressurized clean utility systems, leakage of pure water or sterile air into the surrounding environment normally will not be hazardous; however, it can be expensive, in terms of lost fluids and clean up.
Leakage of pure steam out of the system can present a safety issue as well as an expense. Leakage into the steam system creates an even worse situation. For example, clean steam is used for sterilization. After the necessary temperatures are reached, stabilized, and held for the prescribed length of time, the equipment or system is cooled.
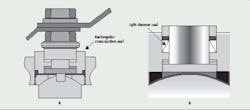
During cool down, a vacuum is created inside, and if the stem seals in ball valves used for isolation have failed, contaminating microorganisms may be drawn into the system. Generally, ball valve stem seals consist of a ring of polytetrafluoroethylene (PTFE) having a square or rectangular cross-section and are contained on the outside diameter (OD) by the packing bore wall, on the inside diameter (ID) by the valve stem, and on the top and bottom by washer-shaped glands (Figure 1a).
Figure 1 (A and B): Figure 1A shows a ball valve stem seal that consists of a ring of PTFE with a rectangular cross-section. Figure 1B illustrates the
live-loaded, 2-piece chevron stem packing, which requires less operating torque, improves performance, and compensates for stem wear.
Most manufacturers choose PTFE for stem seals. It is relatively easy to deform the material to make an initial seal. However, it can cold flow, or continue to deform under load—a condition which worsens with increasing temperature. Because PTFE has no “memory,” once it is deformed under load, it does not return to its original shape when the load is removed.
The seal is made by deforming the seal ring by applying sufficient force through a packing nut to deform the material inward against the stem and outward against the packing bore. It is necessary to fully encapsulate the PTFE seal material on all surfaces with metal—the packing bore, valve stem, and glands—as described. The clearance between the gland ID and the valve stem OD should be an absolute minimum.
Otherwise, during thermal cycling, when the temperature of the valve is increased from ambient up to the sterilization point and back down, the material can migrate or cold flow out of the seal area, eventually loosening the seal. In addition, the stem seal must maintain its integrity during the rotation of the stem within the PTFE seal member. Surface finish on the valve stem is important in terms of reducing wear on the inside diameter of the seal.
ASME BPE addresses requirements for rotating valve stem seals but does not specify surface finish requirements for the stem. The metal surface of the stem will have a certain level of roughness, in the form of “peaks and valleys,” from the machining operations. Under load and with thermal cycling, the PTFE seal material cold flows into the valleys.
Then, when the valve is actuated, the small amounts of material in the valleys are sheared off and migrate out of the seal area. As the valve is cycled, more material is removed, the initial load that made up the seal is reduced, and the seal becomes loose. Both of these situations result in rapid wear of the seal, leading to potential leakage.
The degree of encapsulation of the PTFE seal in various ball valve designs and the manufactured surface finish on the stems in valves from different manufacturers impact how quickly stem seal failure might occur—in many cases after only a few thermal cycles. If suitable head pressure is not maintained in the system or equipment during cool down from sterilization temperatures, unwanted microorganisms can be drawn in and destroy the sterile condition.
Although pressurizing with sterile air can help avoid this situation, these microorganisms can still migrate across the loosened stem seal and contaminate the process. General improvements to ball valve stem seal reliability can be accomplished by improving the containment, or encapsulation, of the seal member and by improving the seal surface on the stem during manufacture. Further improvements in seal reliability have been made by improving the typical configuration of the seal member and by adding a live-loading mechanism.
Liveloading means that as the stem seal wears, sufficient load is consistently applied to maintain the seal. One approach to live-loading uses a seal ring with a two-piece split chevron configuration (Figure 1b), rather than the typical one-piece square or rectangular crosssection. The two angled, conical pieces of the chevron create a wedging action to achieve a seal against both the packing bore and the stem. The force required to initially make-up this seal is lower than the force needed to make a seal with a solid, one-piece packing.
A group of conical disc springs placed on top of the seal member provide the live-loading force and compensate for expansion and contraction during thermal cycling. As the seal wears during normal use, the springs continually “retighten” the seal, ensuring its integrity and reliability over a longer service life.
Seat Seals
Ball valve seats are also dynamic seals, which function as the valve is cycled open and closed. Most ball valves are designed with floating balls.
That is, when the valve is in the closed position, the ball is free to move axially toward the downstream seat under the action of upstream pressure. In floating ball designs, the upstream pressure is normally required to effect and maintain shutoff. ASME BPE specifically requires that ball valve closure members (seats) must not be pressure dependent.
In utility systems, there are applications where upstream pressure isn’t adequate to achieve the required level of leak-tight shut-off. Seats in most ball valves are also made from PTFE and, therefore, have similar requirements as the stem seal. For example, the same kinds of requirements for surface finish on the seal surface, in this case the ball, and containment of the PTFE seal member, in this case the seat, apply. Generally, surface finish on the ball is not a problem.
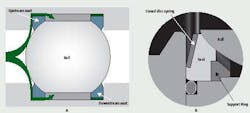
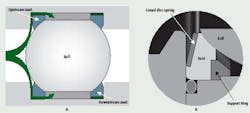
Most manufacturers use spherically ground balls, and unless the surface is somehow damaged, the surfaces are satisfactory. The seats, however, can be a problem. The shape of the seat in most ball valves is a ring, generally a square or rectangular cross-section, with the surface facing the ball contoured to mate with it (Figure2a). In most designs, the seat is not contained on the inside or outside diameters, leaving plenty of room for the seal member to cold flow under normal loads or extrude under higher loads.
As long as there is sufficient upstream pressure, the valves should continue to function. However, at some point the seats can become so distorted that shutoff cannot be achieved and internal seat leakage is a possibility. The comments regarding both thermal and mechanical cycling also apply, though thermal cycling represents the larger problem.
With the outside and inside diameters of the seal member unconstrained, the downstream seat can be permanently deformed, allowing the ball to move further and further into the downstream seat and away from the upstream seat. If the upstream seat is not also sealed against the ball, steam can condense behind the ball. When cooled, contamination can build up. The downstream seat may become sufficiently distorted such that in the open position, where the ball is not free to float, the ball may not be in contact with that seat.
This situation creates a path to permit the contamination to be drawn into the process stream. As with the stem seal, containment of the seat is important in order to resist the possible distortion of the seat that can be caused by temperature and pressure. In addition, live loading the seat provides some real improvements in both downstream and upstream sealing on the ball.
Live loading the seats with conical disc springs ensures that the seats maintain contact with the surface of the ball at all times, making a leak-tight seal on both the upstream and downstream sides of the ball (Figure 2b). With the valve in the open position, a null point is established, and both the upstream and downstream seats are sealed against the ball. In the closed position, as the backpressure is increased, the ball will move toward the downstream seat.
Figure 2 (A and B): The ball valve seat in Figure 2A is not contained. The coned-disc, spring-loaded seat, shown in Figure 2B, ensures a leak-tight seal
on both the upstream and downstream sides of the ball.
During this action, the conical disc spring supporting the seat flexes and permits the seat to move without distortion, while maintaining the seal against the ball. In this arrangement, the seats do not require backpressure for leak-tight shut-off, so the valves may be used effectively in low pressure and mild vacuum service.
The live-loaded seats compensate for expansion and contraction of the components during thermal cycling. This compensating action resists the potential for overloading the seats and causing the severe distortion that leads to internal leakage across the seat.