Migrating from Vials to Prefilled Injectables
A pharmaceutical industry trend is the migration from vials to prefilled syringes especially for biologics. Since this is the final administered dose, fill control requirements are rigorous and leading pharmaceutical producers rely heavily on manufacturing analytics (MA) for control.
Risk reduction, cost control and more accurate drug delivery drive the migration from vials to prefilled injectables:
- Overfill reduction
o Eliminates the need to overfill vials to ensure adequate dose delivery
o Eliminate fraud due to retrieval of left-over medication
o Reduce costs especially for expensive biologics
o Increase dose yield per batch - Risk reduction
o For the manufacturer – individually packaged doses reduce chance of contamination and degradation
o For the medical practitioner – reduce chance of administering wrong dose
o For the patient – more accurate dose for self-administered pharmaceuticals
While inspection is necessary, real process control depends on MA, specifically statistical process control (SPC) methods. SPC is essential to actually delivering on the promise of greater compliance and better control of delivered doses while minimizing syringe production failure and production disruption.
High performance SPC for high performance production systems
Life sciences companies try to increase throughput while maintaining process stability, product quality and compliance. One common solution for prefilled injectables is to deploy equipment which simultaneously fills multiple syringes with each cycle.
Multi-head syringe filling equipment fits the definition of a “family” process where individual sub-processes are constituent parts of a global process. Conventional SPC methods do not deal effectively with family processes and the median-individual (M/I) chart was developed to efficiently monitor and manage family processes such as syringe filling systems.
Family processes share many or all the following characteristics:
| Characteristic | Example |
| The “family” consists of multiple similar or identical member processes originating from a common source or manufacturing stream. | Syringe filling machines typically use multiple fill heads. |
| The member processes are completed more or less simultaneously. | All syringes are filled in a single step. |
| Each member process may have its own mechanical component(s) or subsystem. | Each syringe filler head has its own peristaltic pump and fill orifice. |
| Each member process has its own sources of variation. | Syringe filler head performance can vary independently. |
| The family process as a whole is subject to its own global causes of variation. | Syringe fill line pressure or material viscosity may fluctuate. |
NWA has led the development and implementation of the M/I methodology. The white paper Median/Individual Measurements Control Charting and Analysis for Family Processes details the method and its application. Multi-head filling systems such as syringe filling are one of the most common application areas. Using M/I analytics fits the intent of PAT and gives individual pharmaceutical firms the chance for competitive advantage while developing higher compliance.
M/I charting to monitor syringe fill
Several leading pharmaceutical firms have adopted the M/I methods to monitor and control their filling operations for vials, syringes and capsules. Tight fill process control is especially critical for syringes since that fill is the final administered dose.
Family processes such as syringe filling machinery are widely used because they substantially increase the throughput and operational convenience of a manufacturing line. However, there are real problems monitoring and controlling family processes with conventional charting methods.
Family processes are affected by two types of factors: those that influence all family members and those that influence only the individual member. Since control charts such as X-Bar show only global sources of variation, they cannot pinpoint local assignable causes for individual family members.
For example, consider the results when a single-fill channel is blocked in a ten orifice filler. An X-Bar control chart uses a random sampling from all cavities and therefore cannot identify the specific cavity with the obstructed channel. The channel may remain blocked, yet the chart will show the overall process to be in statistical control. Conversely, charting each orifice—each family member—would be impractical and time-consuming.
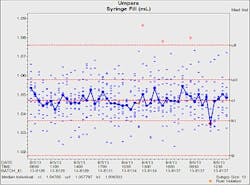
In the process charted in figure 1, a ten head system is filling syringes with a nominal 1 mL dose. The solid blue line represents the global fill process and the blue characters represent the performance of individual heads. The individual values in control are plotted anonymously. Out of control values are plotted in red and the individual heads identified.
Figure 1 – M/I chart monitors 1 mL syringe filler
In this case the individuals plot shows an overfill problem on filler orifice 9 on August 4 which directs engineering to correct the problem. The median line representing the global process exhibits two signals, a rule violation on August 4 and an out of control signal on the 5th. The process was restabilized rapidly. Fortunately, the out of control fill head did not produce out of specification product.
Additional supporting analytics
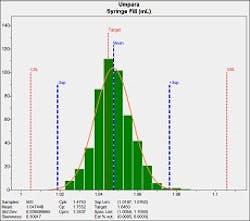
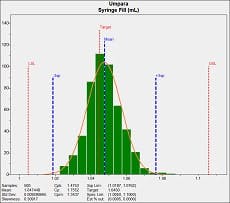
As can be seen in the process capability report (figure 2), the filling process is nominally capable with a Cpk of 1.4753 and no out of conformance product was produced. In critical processes such as producing prefilled injectables, it is best practice to increase the process capability as much as possible to virtually eliminate the likelihood of producing out of compliance syringes. This involves either shifting the mean value or reducing process standard deviation. Drug administration safety is a compelling reason for high capability manufacturing.
Figure 2 – Process capability analysis of syringe filler
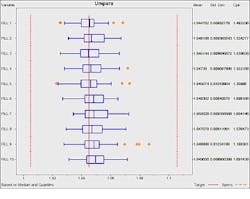
Other analytics techniques such as exploratory data analysis provide additional insights into the process performance. Box plots, for example, provide an alternate means to view process variation compared to histograms.
With box plots engineering can quickly and easily review behavior of both the overall process and the individual heads. In figure 3, the box plot shows the behavior of each fill orifice graphically along with selected statistics. The outliers from fill head 9 are clearly evident
Figure 3 – Box plot of syringe filler head performance
MA - the means to high capability production and compliance
Prefilled injectables represent the state of the art technology to administer pharmaceuticals and biologics. Since the syringes represent the final administered dose, the fill is critical and the process needs to be tightly controlled.
Manufacturing Analytics is the state of the art methodology to monitor and improve pharmaceutical production. Advanced SPC methods such as M/I charting enable pharmaceutical manufacturers to maximize compliance and process efficiency while reducing risk and cost.