The Pharmaceutical Manufacturing editorial team recently queried its readers for the third time in an effort to understand more clearly how CapEx is being managed where they work, and the spending priorities that support their operational strategies.
STILL RISING
The pharmaceutical industry has reported capex spending to be on the rise since 2013. There has been a significant increase in equipment budgets from 2013 to 2015 — in fact, a whopping 69 percent, according to the Nice Insight’s 2016 Pharmaceutical Equipment Annual Survey of 489 global pharmaceutical and biotech industry professionals involved in purchasing new systems and technology.
Pharmaceutical Manufacturing’s surveyed readers indicated that the trend has continued in 2016. Almost 63 percent of surveyed readers report that from FY 2015 to FY 2016, capital spending on production assets/technologies and process systems at their companies is increasing. Only 17 percent reported a decrease in spending.
Specifically, when readers were asked to rate their company’s capital spending/investment performance over the last year in terms of improving production infrastructure, more than 50 percent described it as “cautious, but adequate, keeping pace with professed productivity, safety and quality-associated initiatives.” Twenty-four percent gauged it to be “ample and a high priority, focused on meeting present and future business goals.”
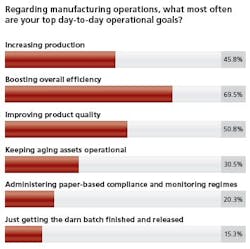
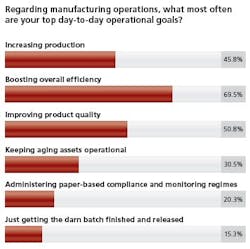
In their quest to achieve these day-to-day goals, more than 86 percent feel process innovation and improvement is important or very important to their organization.
THE CURRENT SITUATION
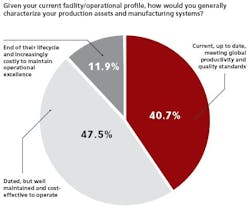
When asked how they would characterize their company’s production assets and manufacturing systems, readers generally had good news to report. More than 40 percent reported assets and systems to be up-to-date, meeting global productivity and quality standards; and 47.5 percent say they are dated, but yet well-maintained and cost effective to operate.
So when it’s time to invest, on what sort of shop floor systems are companies spending the most capital dollars? Higher speed, higher-capacity processing systems and equipment to meet increasing product demand or new product production is in the lead (35.6 percent), followed by adding process analytical technologies to existing lines to understand critical processes better (23.7 percent).
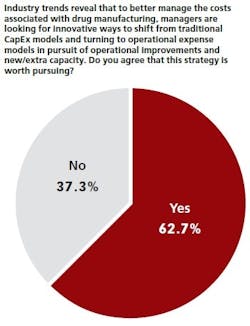
OPEX SHIFT
Those who agree this strategy is worth pursuing are applying their manufacturing-related OpEx dollars mostly toward IT systems/infrastructure (48.6 percent) and late-stage development production (40 percent).
WHO ANSWERED
Representing manufacturing’s most important roles, the survey attracted individuals from all operational levels —from executive leadership to plant floor operatives. Of all respondents, nearly 83 percent have responsibilities that include supporting internal processes associated with the specification/purchase of process and production technologies. The survey found that 66 percent of respondents have spending authority in their current roles. More than 42 percent are authorized to spend more than $100,000.
About the Author
Karen P. Langhauser
Chief Content Director, Pharma Manufacturing
Karen currently serves as Pharma Manufacturing's chief content director.
Now having dedicated her entire career to b2b journalism, Karen got her start writing for Food Manufacturing magazine. She made the decision to trade food for drugs in 2013, when she joined Putman Media as the digital content manager for Pharma Manufacturing, later taking the helm on the brand in 2016.
As an award-winning journalist with 20+ years experience writing in the manufacturing space, Karen passionately believes that b2b content does not have to suck. As the content director, her ongoing mission has been to keep Pharma Manufacturing's editorial look, tone and content fresh and accessible.
Karen graduated with honors from Bucknell University, where she majored in English and played Division 1 softball for the Bison. Happily living in NJ's famed Asbury Park, Karen is a retired Garden State Rollergirl, known to the roller derby community as the 'Predator-in-Chief.'