An ongoing pattern of hesitancy exists within the pharmaceutical industry, a reticence that highlights how slow companies are to adopt new technology—an unfortunate trend that is costing companies literally billions of dollars every year. New data indicates that when it comes to Enterprise Quality Management Software (EQMS) implementation, managers are being particularly hesitant to move forward. According to LNS Research in its recently published Research Spotlight “The Cost of Inaction - Taking Quality Management Processes Digital” the industry’s uncertainty about adopting useful technologies like EQMS stems from the same issue: ”While nearly all executives acknowledge that quality is at the core of most business challenges, many position the health and capability of their quality management solution far down on the list of priorities.”
Unfortunately, the gap between quality needs and actual EQMS investment is nothing new. In 2008 Nora Dyer, Global Head of Clinical Development Operations at Novartis, suggested that a lack of understanding among key decision makers is the biggest hurdle to adopting an automated solution. “Adoption of e-solutions is hindered when important stakeholders do not clearly understand and support the overall business rationale,” said Dyer, “and don’t drive towards the change. Therefore, an effective communication strategy using multiple channels to reach the end-user is essential for a successful implementation.” 1
Most quality professionals see the need for an EQMS system. But the main challenge today, as in 2008, seems to be focused on executives and the budgets required to implement an EQMS system. As the data from 2008 and the more current data from LNS indicates, companies’ budgets are still being spent on what are perceived to be higher, more immediate priorities. Yet, improperly prioritizing EQMS can cost a pharmaceutical company millions of dollars each year, as well as cause them to lose market share to competitors who have already successfully invested in an EQMS system.
LNS surveyed over 750 quality management executives and senior leaders. The results of this survey were telling. While more companies are talking about adopting an EQMS system, few are making progress when it comes to actually budgeting for and implementing such a system.
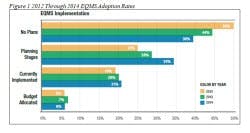
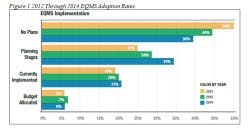
In Figure 1, Survey results show a clear trend when it comes to investing in EQMS. As you look at the graph, note the following observations:
1. Fewer companies each year say they have “no plans” to implement an EQMS system. This indicates that it’s on their minds more than before.
2. More and more companies are in the planning stages of implementing an EQMS – up from 25 percent in 2012 to 34 percent in 2014.
3. The number of companies that have actually implemented an EQMS has remained more or less steady, with conservative growth from 19 percent in 2012 to only 21 percent in 2014.
4. Actual budgets allocated to adopting an EQMS have remained stagnant at 6 percent to 7 percent over the last three years.
Not surprisingly, this corresponds with data MasterControl recently collected from almost 300 qualified prospects. Of those companies that fell out of the sales pipeline, over half declared either “no budget” or “other priorities” as the main reason for not moving forward with an EQMS. (Note that all of these companies were pre-qualified prospects who actively expressed an interest in getting an EQMS for their organization.) Just as the data from LNS indicates, even though quality management is viewed as being crucial, executives are still reluctant to invest in automated systems that can vastly improve this process, reduce costs, and increase profits.
The conclusion MasterControl draws from this data is that many executives managing highly regulated companies are still hesitating to implement EQMS system, especially compared to other departments that are eventually allocated the budget instead. It’s fair to say that most of these companies have the budget for an EQMS but are simply choosing to spend it on other priorities.
THE REAL EQMS ROI
LNS’ research indicates that, “…those companies with closed-loop quality processes recorded a median OEE (Overall Equipment Effectiveness) of 90 percent, which is 10 percent higher than those without closed-loop processes. Additionally, companies with closed-loop processes recorded a 4-percent higher median product compliance and 2 percent higher performance in percentage of on-time deliveries.”
Stating ROI another way, those companies who have implemented an EQMS improve manufacturing efficiency by 20 percent (from 80 to 90 percent), experience less than 1 percent product incompliance, and deliver 97 percent of their products on time. It doesn’t take a lot of accounting prowess to show that for even an average-sized, regulated company, these improvements can add up to millions of dollars in both cost reduction and additional revenue generated.
LNS points out that if you’re one of the 80 percent of regulated companies that have not yet implemented an EQMS system, the other 20 percent (your competitors) are probably experiencing a significant competitive advantage over you already. The “enemy” to overcoming this competitive disadvantage is the inability to replace outdated (and perhaps home-grown) quality systems with a proven, commercial EQMS system. According to LNS, “While homegrown and point solutions deliver value up to some extent, many maturing organizations have surpassed their usefulness and are essentially forcing quality management professionals to ensure quality at the global scale with less resources than competitors. Whether executives are facing pressures to improve costs, achieve compliance, serve customers better, or introduce products to the market faster, there is a strong connection between each of those goals and quality management. And next-generation quality management technology is accelerating progress toward those goals.”
For smaller regulated companies, investing in an EQMS system may spell the difference between one company’s ability to achieve its business and financial goals and being pushed out of business entirely.
WHAT'S TAKING SO LONG?
Pharmaceutical companies are making progress in understanding the need for a functionally efficient EQMS system, but making relatively little progress in actually implementing the technology. Consequently, companies are losing ground to competitors, often without even realizing it.
Most quality professionals already see the value of implementing an EQMS system. It’s the executives that must eventually begin to recognize the ROI to be achieved by properly implementing EQMS into their operations. To help the effort, LNS recommends executives proactively pursue the following five steps:
1. Execute a quality gap analysis: Develop a cross-functional team to inventory current quality systems, data sources, and processes (NC/CAPA, audit management, supplier quality management, risk management, document management etc.) across the value chain. Use the same team to develop a list of ideal requirements for world-class quality management. Compare current versus ideal to execute a quality gap analysis.
2. Build a business case for an EQMS investment: Leverage benchmark data on operational and financial performance improvements experienced by organizations with EQMS versus those without EQMS. Use performance data on metrics such as OEE, percent of products in compliance, on-time deliveries, and rate of new product introductions to quantify an ROI and build a business case.
3. Evaluate EQMS solutions providers: Take a comparative approach to researching the EQMS solutions vendor market and develop a future vision matching ideal requirements. Evaluate each vendor with a live demo. Consider vendors able to serve current ideal requirements, but also those investing in emerging technology trends such as big data analytics.
4. Implement EQMS functionalities and prove value: Some organizations may find it optimal to deploy entire EQMS suites of functionality, while others may have budget for only specific functionalities and choose to implement modularly. After making a solution selection, deploy EQMS functionalities as business needs permit.
5. Scale EQMS investment as needed/budget permits: An EQMS investment may start in the manufacturing, engineering, or corporate quality environment, but will generally extend out into other areas of the value chain over time. Always deploy functionalities with the future vision of closed-loop quality processes in mind.
The bottom line for pharmaceutical companies trying to compete in today’s high-tech world is that adopting new software technologies is no longer a luxury or superfluous to a company’s success—but, rather, an essential key to success in an industry that’s becoming increasingly competitive. Those who adopt this view are far more likely to succeed than those who do not.
¹Dyer, N., as quoted by Christel, M.D. (2007, July/August). E-Solutions in Clinical Trials: Digital Dilemma. R&D Directions. Retrieved September 12, 2007, from http://www.pharmalive.com.